It is well known that a complex signals network involving immune and neuroendocrine systems plays a critical role in the modulation of inflammatory and immune responses against environmental danger (e.g. pathogens, nutritional and/or environmental factors) that involve a systemic response.
The aim is the maintenance of homeostasis, the dynamic equilibrium in which continuous changes set the optimal conditions for survival. Multi-sector communication guarantees the maintenance of homeostasis and therefore of health.

The immune, endocrine and central nervous systems communicate through anatomical and hormonal-neuropeptides pathways.
The efferent nerves of the sympathetic nervous system innervate primary and secondary lymphoid organs; adrenaline and noradrenaline have been shown to activate the hypothalamus-pituitary-adrenal (HPA) axis and to act on leucocyte adrenergic β2 receptors.
Immune cells express receptors for many hormones (e.g adrenocorticotropic hormone, prolactin, growth hormone (GH), leptin, glucocorticoids, estrogens and testosterone, neurotransmitters and neuropeptides such as VIP, substance P, norepinephrine, somatostatin and neuropeptide Y). Equally, endocrine and nervous tissues show specific receptors for immune-derived cytokines.
Within this framework, it is also clear that immune cell functions can be altered by neuroendocrine hormones and that immune cells have the ability to produce hormones with a functional neuroendocrine response.
Hormones and cytokines, particularly pro-inflammatory and regulatory cytokines, are the main actors of this coordinated cross talk. Their action translates into an enhancement of innate immunity, a control of the inflammation with an increase in the efficiency of the immune response. In fact, it starts also in response to a pathogen challenge to re-establish the homeostasis. Conversely, a dysregulation of this neuro-immune response can negatively affect both immune response against pathogens and metabolic efficiency.
In this “check and balance” system, emerging evidence points to a fundamental role for gut microbiota. Gut microbiota interacts closely with the body’s major neuroendocrine system that controls various body processes in response to stress, the hypothalamic–pituitary–adrenal (HPA) axis. This interaction is of pivotal importance as various disorders of the microbiota–gut–brain axis can be associated with dysregulation of the HPA axis and of the inter-correlated immune system.
The activation of the HPA axis and secretion of glucocorticoids is one of the major neuroendocrine pathways to control local and systemic inflammation.
However, prolactin and GH are also involved in the development and efficiency of the immune response. They are signals that facilitate the proliferation, differentiation and function of immunocompetent cells towards specific antigenic stimuli by an endocrine and paracrine action. In fact, both hormones, which are identical to the hormones of the endocrine glands, are synthesized, stored and secreted by immune cells.
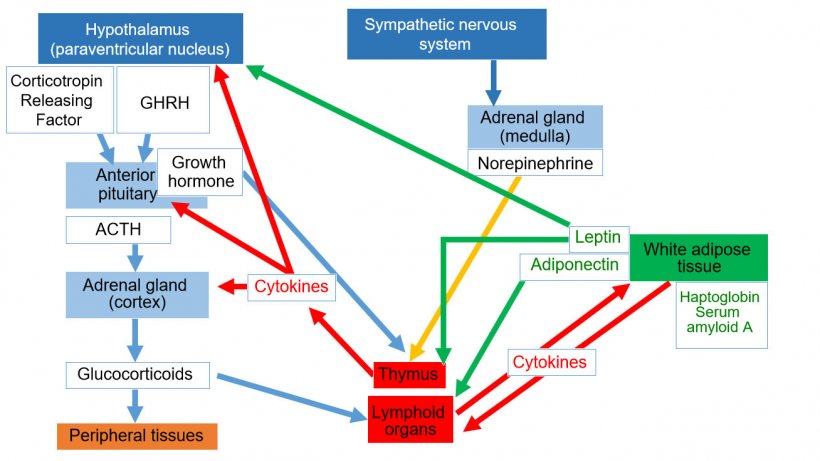
White Adipose Tissue plays a pivotal role in the neuro-immunity network linking nutritional status, metabolism and energy balance to maintain body homeostasis. Products primarily synthesized in adipose tissue are known as adipokines including anti- and pro-inflammatory factors such as classical cytokines, but also specific hormones. The main hormones, leptin and adiponectin, have also been identified as an important players in the crosstalk between endocrine and immune system.
Leptin not only regulates food intake and metabolism, but also stimulates the proliferation and survival of lymphoid precursor and the development of T cells in the thymus. It can also improve the Th1 response by increasing IL-2 and IFN-ϒ production by T cells and macrophages.
Adiponectin includes multiple isoforms and is the most highly expressed adipokine. High molecular weight adiponectin acts primarily as an anti-inflammatory regulator through suppression of differentiation and classical activation of M1 macrophages by downregulating pro-inflammatory cytokines, i.e TNF-α and IL-6, acting on adiponectin receptors 1. The lower molecular weight isoforms seem to play a pro-inflammatory role by increasing the activity of pro-inflammatory molecules. (Tong et al., 2011; Gomez et al., 2011) (Figure 1).
In intensive pig breeding, several stressors such as management practices, environmental stressors, nutritional factors, weaning, mixing, fighting, shipping can negatively influence the immune responses in terms of functional efficiency.

It is evident that the first weeks of life constitute the most critical period for piglets because the development of the immune system and its efficiency increase progressively. Indeed, during the neonatal period, piglets can be exposed to physical and psychological stressors and infections that can directly influence the efficiency of immune response.
Weaning is one of the most stressful events that can predispose to intestinal and immune system dysfunctions, severely compromising health, growth, feed intake, particularly during the first week after weaning.
Similarly, infections are major contributing factors associated with reduced performance in food animals during growth. Persistent/subclinical infections can also negatively affect the growth performance influencing the hormonal axis, tissue metabolism and nutritional efficiency.
During infection, the activation of the HPA axis and secretion of glucocorticoids can influence both the severity and the metabolic impact of the infection.
Indeed, an uncontrolled inflammation, with potential tissue damage, depends on a complex interaction between glucocorticoid levels, their peripheral activity and, mostly, the levels and persistence of pro-inflammatory cytokines (TNF-a, IL-1ϐ, IL-6, IL-15).
The HPA axis and GH-insulin-like growth factor 1 (IGF1) axis activation/dysregulation and altered systemic cytokine levels may induce metabolic changes. At the beginning of infection/inflammation, several metabolic changes (GH rise) can support an efficient innate immune response (energy switched towards immune system activation). Thereafter, an uncoupling between GH and IGF-1 levels occurs with GH axis suppression, liver and skeletal GH resistance and inhibition of IGF-1 activity on peripheral tissues (Soendergaard et al., 2017). In addition, high levels of glucocorticoids and pro-inflammatory cytokines (i.e. IL-1, TNF-α and IL-6) induce a reduction of food intake, a negative energy balance with tissue catabolism, protein and aminoacids breakdown and reduced bioavailability and secretion of oligo-elements (e.g, Zn and Cu).
Figure 2: Effects of inflammation on reduced performances and growth.
Table 1. Main signal of immune-neuroendocrine communication
| Hormones | Glucocorticoids |
|
|
Growth Hormone (GH) |
|
|
| Leptin |
|
|
| Adiponectin |
|
|
| Cytokines | IL-1, TNFa, IL-6 |
|





















