Enterotoxigenic Escherichia coli (ETEC) remains a major cause of illness and death in neonatal and recently weaned pigs. Pigs that develop diarrhoea after weaning (post-weaning diarrhea; PWD) do so generally within 3-10 days. Strains of ETEC that cause PWD possess two types of virulence factors, adhesins and enterotoxins, both of which are essential for PWD to express itself clinically. Porcine ETEC isolates produce any of five different adhesins, all of which are fimbriae (or pili): K88 (F4), K99 (F5), 987P (F6), F41 (F7) and F18. Fimbrial adhesins K88 and F18 occur in several antigenic forms, with the commonly occurring K88 fimbrial variants including K88ab, K88ac, and K88ad. Fimbrial adhesins target specific receptors on brush border enterocytes of the small intestine that then enable the bacteria to colonise the cell surface and excrete toxins, with the predominant toxins produced by ETEC strains causing PWD being heat labile enterotoxin (LT), heat stable enterotoxin type A (STa), heat stable enterotoxin type B (STb), and (or) enteroaggregative E. coli heat-stable enterotoxin1 (EAST1). Enterotoxin Stx2e (Shiga toxin), also known as oedema disease factor, is the cause of lesions associated with oedema disease in pigs (see Table 1, for summary).
Interestingly, the B subunit of LT (LTB) has immunomodulatory properties from which can arise apoptosis (cell death) of CD8-positive T cells, activation of B cells, and alteration of cytokine secretion by the monocytes. The adjuvant properties of LTB were demonstrated in a study showing superior protection against ETEC PWD in weaned pigs by a live E. coli vaccine that had the genes for LTB and K88ac compared with an isogenic strain that lacked the gene for LTB. The strain that had LTB alone was not effective in eliciting protective immunity. In contrast, toxins STa and STb are small molecules and considered poorly immunogenic, and these antigens alone cannot induce pig anti-STa and anti-STb immune responses that are required for effective protection against PWD.

Table 1. A summary of virulence factors of ETEC associated with PWD (reproduced from Zhang, 2014).
| I. Adhesins | References | |
| Fimbrial adhesins | Morphology (diameter) & structural subunit (kDa) | |
| K88 (F4) | Fibrillar (2.1 nm), FaeGa(27.6)/FaeCb(16.9) | Bakker et al., 1991 |
| K99 (F5) | Fibrillar (4.8 nm), FanCa(16.5)/FanFb(31.5) | Isaacson, 1977; Bakker et al., 1991 |
| 987P (F6) | Fimbrial (7.0 nm), FasAa(23.0)/FasFb(17.5)/FasGb(40) | Isaacson & Ritcher, 1981; Edwards et al., 1996 |
| F41 (F7) | Fibrillar (3.2 nm), Fim41aa(29.5) | deGraaf, 1982 |
| F18 (F107; 2134P; CFA8813) | Fibrillar (3-4 nm), FedAa(15.1)/FedFb(30.3) | Imberechts et al., 1996; Nagy et al., 1997 #17795; Smeds et al., 2001 |
| Non-fimbrial adhesins | ||
| AIDA-I | Autotransporter protein, AidA (79.5c) | Benz & Schmidt, 1992; Benz & Schmidt, 2001 |
| ppa | Outermembrane protein, Paa (27.6) | Batisson et al., 2003 |
| EAE | Outermembrane protein, EaeA (94-97) | Jerse & Krapper, 1991 |
| II. Toxins | Gene, mature toxin protein | |
| LT | eltAB, 1:5 AB holotoxin (86 kDa) | Sixma et al., 1991; Streatfield, 1992 |
| STa | estA, 18 amino acid peptide (2kDa) | Dreyfus et al., 1983 |
| STb | estB, 48 amino acid peptide (5.1 kDa) | Lee et al., 1983; Dreyfus et al., 1992 |
| EAST1 | astA, 38 amino acid peptide (4.1 kDa) | Nataro et al., 1987 |
| Stx2e | stx2e, 1:5 AB toxin (70 kDa) | Pierard et al., 1991 |
a: major structural subunit
b: minor structural and/or fimbrial tip subunit
c: mature AIDA-I adhesin is detected as 100 kDa by SDS-PAGE
Not all ETEC strains possess F4 fimbria and this explains, in part, some of the variation seen between farms in PWD. Nevertheless, vaccination of a sow during pregnancy leads to the secretion of antigen-specific antibodies in colostrum and milk, which can protect pre-weaned piglets against ETEC infection. Weaning, however, causes the loss of milk antibodies and IgG that appear to contribute to the enhanced susceptibility of pigs to E. coli enteric infections after weaning, and subsequently an active mucosal immune response is needed to provide protection. Specifically, activation of the mucosal immune system of the small intestine, specifically the jejunal Peyer’s patches, to evoke F4 antigen-specific IgA or IgM responses is required to induce a protective mucosal immune response. As seen from Figure 1, however, it takes some time after weaning for IgA levels to increase in response to any pathogenic challenge.
Since ETEC strains are by far the most common and important cause of PWD, this group of pathogenic E. coli has been primarily targeted in vaccine development. Vaccine developments against PWD have been on-going for many years, however and as explained by Zhang (2014), there are challenges in developing cost effective vaccines against PWD. Enterotoxins are virulence determinants in PWD and so must be included as vaccine components even when LT, STa and STb are potently toxic and cannot be used as safe antigens. Hence, an effective PWD vaccine would have to induce anti-adhesin immunity against K88 and (or) F18 fimbriae and also antitoxin immunity at least against LT, STa and STb. These fimbriae and toxins are genetically and immunologically heterogeneous and therefore, immunity induced by one fimbria or toxin provides protection only against ETEC strains expressing the same fimbria or toxin, but cannot cross protect against ETEC strains expressing a different fimbria or a different toxin.
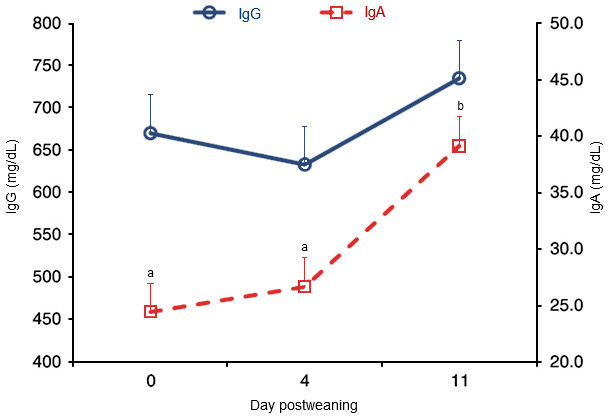
Figure 1. Concentrations of plasma IgG and IgA (mg/dL) on day 0, 4, and 11 after weaning. The concentration of IgA was higher (P < 0.001) on day 11 following weaning than on day 0 or 4 following weaning [Pigs were weaned on day 0 and challenged with E. coli F18 on day 1, 2, and 3 after weaning. Plasma was collected on day 0 before the piglets were moved to the nursery pen] (from Sugaharto et al., 2014).
In addition, pigs of various genetic backgrounds express different receptors, and these receptors are recognised and adhered by specific fimbriae expressed by different ETEC strains. If orally immunised with a fimbrial vaccine product, only pigs expressing receptors specific to this fimbria will effectively develop immune responses. Even so, an effective PWD vaccine should induce protective anti-adhesin immunity against K88 and (or) F18 fimbriae but also antitoxin immunity against LT and ST toxins. A toxoid multi epitope fusion antigen (MEFA) strategy (involving neutralizing antibodies against toxins) has allowed multiple adhesion and toxin antigens to be potentially included into a single product for development of safe and truly broad-spectrum protective PWD vaccines, however further experimentation is required before market release.
In practice, and to have active-acquired antibodies ready at weaning, piglets should be immunised 10 days to 2 weeks before weaning. However, the required level of passive maternal antibodies in suckling piglets must be high enough to protect against neonatal diarrhoea but low enough so that these antibodies do not compromise the immunised antigens in their role in inducing specific antibodies against PWD. Vaccinating individual pigs or live oral immunisation at weaning (e.g., adding vaccine products in feeds or drinking water) are means by which immunity can be delivered.






