Porcine circovirus type 2 (PCV2) is linked to several clinical disease manifestations in all stages of pig production commonly referred to as PCV-associated diseases (PCVAD). Infected pigs shed PCV2 in essentially all body secretions and excretions and horizontal transmission is of great importance. PCV2 infection in growing pig has been well studied under experimental conditions and in the field; however, still many questions remain on the importance of PCV2 infection in breeding herds. The following is a summary of current knowledge to address frequently asked questions from producers and practitioners on PCV2-associated reproductive disease:
Is PCV2 associated reproductive failure important and how prevalent is it?
Breeding herds may be directly affected by PCV2 through infection of embryos and fetuses during dam viremia resulting in early embryonic death, abortion, or decreased reproductive performances manifest as decreased litter sizes and increased numbers of mummified fetuses (Fig. 1). PCV2-associated reproductive failure is observed in PCV2 negative or naïve pregnant dams after waning of passively derived antibodies. As PCV2 is widespread, with the possible exception of gilt start-up herds, the numbers of susceptible animals within a herd is generally thought to be low, and therefore, PCV2-associated reproductive failure is often hard to recognize clinically because of the low numbers of affected animals.

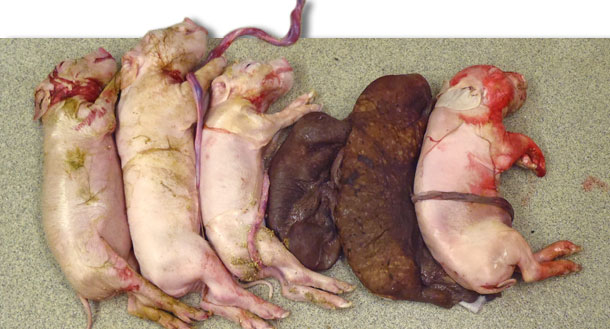
Fig 1. Litter from a sow experimentally infected with PCV2 at the time of insemination. Note the small litter size and the presence of two mummififed fetuses.
How can PCV2 associated reproductive failure be diagnosed?
PCV2 viremia in the dam may last for multiple weeks during the gestation period and fetuses can be infected at different stages of gestation leading to variably-sized mummified fetuses and stillborns at parturition. An overall increased incidence of mummies may be recognizable; however, once recognized, the affected dams will already have seroconverted to PCV2 and may no longer be viremic. Therefore, diagnostic efforts should focus on the fetuses or piglets. The most accurate tool to diagnose PCV2-associated reproductive failure is demonstration of PCV2 antigen or DNA in fetal tissues and association of this with myocarditis and myocardial fibrosis. As only individual fetuses may be affected, samples (especially heart) need to be collected from several piglets or fetuses within a litter.
Is subclinical PCV2 infection of neonates prevalent?
Our group and others have shown that in otherwise healthy breeding herds, PCV2 viremia prevalence rates in newborn pigs can be as high as 16% to 71%. Based on our experience, it is rather easy to demonstrate presence of PCV2 DNA in neonatal or suckling pig sera.
What does a PCV2 PCR positive result in neonates mean for the pig and for its cohorts?
While clinical PCVAD is most often seen in growing pigs, breeding herds are thought of as contributing to PCV2 survival by infecting very young piglets even before or shortly after birth subsequently resulting in transmission of PCV2 into nurseries and maintenance of PCV2 in this production stage. Viremic piglets, i.e. piglets that are positive for PCV2 DNA in serum, shed infectious PCV2 and are a point source of infection for others. This may particularly be of concern if pigs from different sources are mixed in the nursery, as there is an increased risk for spread and transmission of genetically distinct PCV2 strains. Although PCV2 strains do not differ significantly in virulence under experimental conditions, there is some field evidence indicating that pre-infection of a pig with one strain may result in clinical disease after superinfection with a different strain. In addition, PCV2 is a virus that targets lymphoid tissues thereby interfering with the immune response which could influence the outcome of other virus infections and also the efficacy of routine vaccinations.
What can be done to prevent or reduce PCV2-associated reproductive failure and transmission?
Vaccination of growing pigs against PCV2 has been shown to be highly effective in decreasing the prevalence and severity of PCVAD. Recently, much attention has been given to PCV2 prevention in growing pigs through vaccination of the breeding herd. So far, results have been conflicting as a positive effect of PCV2 vaccination in subclinical PCV2 infected farms is difficult to demonstrate and requires large study populations. In a recent field study our group demonstrated that PCV2 vaccination of sows prior to insemination resulted in a significant reduction of PCV2 viremic pigs at birth. Moreover, under experimental conditions, litters from vaccinated dams had a lower prevalence of intrauterine PCV2 infection with lower PCV2 loads. More work in this area is needed to determine the real benefits of breeding herd vaccination for the breeding herd itself and the offspring.





