Porcine circovirus 2 (PCV2) is causing post-weaning multisystemic wasting syndrome (PMWS) and other PCV2-associated diseases. The potential of PCV2 as a foetal pathogen has been demonstrated by several field reports and experimental studies. PCV2 may reach embryos/foetuses upon transplacental spread during viremia or upon insemination with contaminated semen. The outcome depends on the stage of gestation. Infection of embryos causes embryonic death and return to oestrus in the sow (regular/irregular). Infection of foetuses before 70 days of gestation results in death and mummification. The heart is the main target organ and extensive replication in the myocard cells causes lesions and malfunction (heart failure).
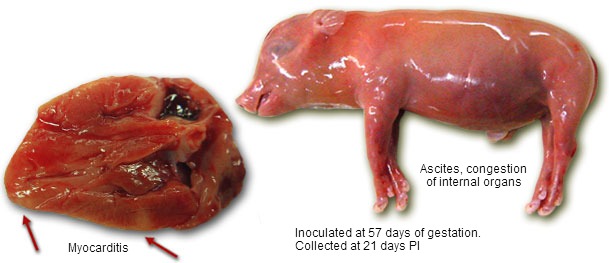

Foetuses after 70 days of gestation have the potential to raise an immune response and the outcome of a PCV2 infection depends on the level of virus replication and the power of the immunity. They may die, survive but be weak and survive without negative effects. In general, PCV2 does not cause abortion but gives affected piglets at birth: mummies and still- and weakborn piglets. Based on all these characteristics, PCV2 has been classified as a SMEDI (Stillbirth, Mummification, Embryonic Death, Infertility) virus. PCV2a and PCV2b strains behave similarly upon intrafetal infection. Due to the exceptionally long replication cycle (36h), it takes time to kill a foetus upon infection and to spread in the uterus.

Seroprevalence studies have shown that PCV2 is widespread in pig populations worldwide and that pigs become systematically infected after losing their maternal immunity. Since most gilts become infected as young pigs, they are immune at their first service/insemination. Therefore, it is difficult to find naïve gilts. As a consequence, the incidence of PCV2-associated reproductive problems is low, as demonstrated by several research groups. However, due to the improvement of their health status, the percentage of naïve gilts is increasing. These susceptible gilts and their embryos/foetuses are considered to be at risk for a PCV2 infection and related reproductive problems. In this context, it is advisable to screen gilts during quarantine before introducing into the sow herd.
Diagnosis of reproductive failure due to an intrauterine PCV2 infection can easily be done by detection of PCV2 (virus isolation/qPCR) or PCV2 infected cells (immunostainings) in the heart, which is the main target in foetuses. Due to the ubiquitous presence of large amounts of PCV2-genetic material in the environment, care should be taken during collecting material for making a PCV2 diagnosis by qPCR. Detecting PCV2 antibodies in foetal fluids to make a diagnosis should be treated with care because low amounts of antibodies may leak through the placenta to foetuses, especially during reproductive problems.
A full proof of the prevention of transplacental spread of PCV2 by vaccination of the sow has not yet been given. However, based on the good control of viremia in pigs by vaccination, this prevention is generally accepted. In this context, vaccination of seronegative gilts is recommended. Vaccination/revaccination of sows may be considered to homogenize their immune status, in order to improve the colostral immunity. A better maternal immunity in piglets will move up the moment of PCV2 infection, away from the moment of infections with other pathogens. Absence of co-infections with PCV2 will improve the clinical situation in the nursery. Reducing co-infecting agents during nursery by a combination of all in/all out measures, effective cleaning/disinfection and antibiotics can further reduce the PCV2-associated problems in pigs.







