The progressive contact with environmental microbes during the first days of life helps the colonization of the gastrointestinal tract (GI) by the bacteria and the establishment of a balance of the GI microbiota. This allows for the development of mucosa associated lymphoid tissue (MALT) that is able to organise an active immune response against harmful microbes but also of tolerating non-harmful antigens as, for instance, commensal bacteria.
A good performance is obtained in the initial stages of production when the environmental conditions do not pose a great challenge for the GI tract barrier, so that more energy is dedicated to growth. This effect was obtained at a low cost with the use of in-feed antibiotics, which could also prevent the transmission of pathogens to the food chain. The effect of antibiotics on the GI microbiota depends on their spectrum, dosage, route of administration and their pharmacokinetic and pharmacodynamic properties. So, with different antibiotics we can attain a growth promoting effect in the piglet through the depressing action exerted on different bacteria.
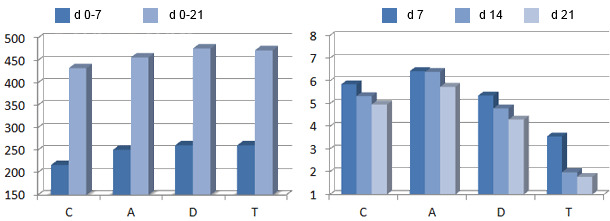
In graphs 1 and 2 we can see how different kinds of antibiotics improved the feed intake in healthy weaned piglets, but hey had different effects on the intestinal microbiota (C = control, A = amoxicillin, D = doxycycline, T = tilmicosin). Through molecular techniques it was seen that the use of these antibiotics caused a reduction of the bacterial diversity regardless of the type of antimicrobial.

 |
|
| Graph 1. Feed intake, g/d. | Graph 2. Enterobacteria count, log10 CFU/g. |
Trevisi et al., 2011
Currently, the EU goals of producing safer animal products include a significant reduction of the hazard that the foods may contain enteric pathogens and antibiotics. Also, a lower use of antibiotics with a therapeutic aim is considered as something that is necessary for reducing the diffusion of antibiotic resistances towards human medicine, and this stimulates the research for finding practical alternatives to the use of antibiotics in pig feeding.
The feeding practices, the choice of the raw materials and the addition of additives have the potential of modulating the GI microbiota, reducing the energy cost for maintenance and also controlling the pathogenic bacteria. Next we will summarise some practical tools for helping to attain this objective:
1- Basic feed formulation. The quality of the polysaccharides in the diet affects the microbiota variability, but it must be synchronised with the protein intake in order to minimise the elimination of protein degradation metabolites. The excess in some nutrients (sugar, iron, etc.) can overstimulate the pathogenic bacteria, and the best solution is to provide highly digestible carbohydrates and few (but good) protein sources.
2- Direct action of the GI microbiota: Organic acids are highly reliable, frequently when combined and partially protected. On the other hand, the essential oils require high doses and are absorbed too quickly in the digestive tract, whilst blood plasma is an interesting but expensive alternative.
3- Enhancement of the intestinal barrier. The maternal environment is the main factor that shapes the GI microbiota of the newborn piglets. The manipulation of the sow's GI microbiota through the diet (including probiotic supplementation) has had a partial success with respect to modifying the piglets' microbiota, their early survival and pre- and post-weaning litter performance. Nevertheless, this strategy has not been adopted by many producers.
During the weaning process, the provision of microbes in the feed can contribute to keep or re-establish the balance in favour of the best commensal bacteria, which suffer a temporary anorexia caused by the weaning stress. So, pig-specific probiotics are required for this purpose as, for instance, some strains of Lactobacillus amylovorus.
Some candidate probiotics have been studied for counteracting some enteric pathogens because of their properties (production of antibacterial molecules, stimulation of the immune response, competition for adhesion sites, etc.). In general, the data show that oral administration can be favourable or at least harmless. Nevertheless, there are cases in which probiotics have been given to pigs that were fighting against a disease, and this has negatively affected the health of these animals. The differences in the results can also be due to the use of different study models or different starting microbiological environments.
There are few reliable data about the specific immune responses of the commensal bacteria and about the interaction of "wild" bacteria with the probiotics administered orally. So, probiotics are not always effective. The tolerance criterion is useful in healthy animals, but it entails a certain risk in animals that are exposed to pathogens. Other probiotics as, for instance, yeasts and some of their products also seem promising with respect to their inclusion in piglets' feed.
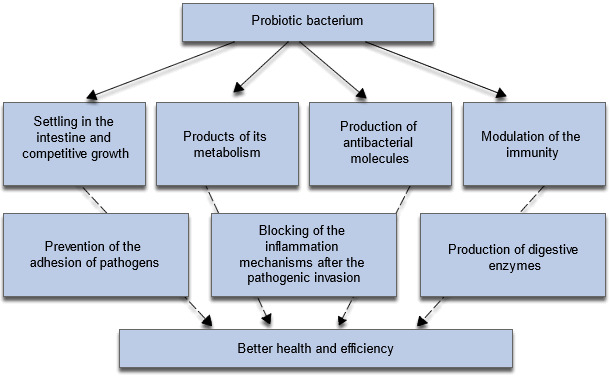
Diagram 3- The mechanisms implied in the positive effects of probiotics on the animals' growth and health.
4- Use of amino acids. Supplementing with tryptophan and threonine can compensate for the increase in the energy cost due to inflammation and the production of mucus and immunoglobulins, especially in pigs that are more susceptible to colibacillosis. Now there are new molecular tests for detecting this individual susceptibility, and this can help us not only in pig selection, but also to improve the feeding practices.
5- Zinc oxide. At pharmaceutical doses it alters significantly the microbiota and can improve growth, but it is not environmentally sustainable.


