Why don't we have a vaccine against ASF yet?
ASF has been a quite forgotten disease, especially if we think in areas in Subsaharian Africa where the virus has remained endemic since its origins, contributing to the poverty and malnutrition in the affected regions. Even when the disease became endemically present in Europe, there were never more than a dozen research groups interested in this topic, with Spain and Portugal being the countries with more researchers implied. Therefore, it is nor strange that in the 60+ years that we know about this virus only 1,454 general articles on ASF or 167 articles on vaccines and ASF have been published.

The first question that we must pose is: Why are we surprised that we do not already have a vaccine against ASF? Prevent to prevent: that should be our motto for the future.
The ASF virus is complex in itself, like its epidemiological cycle, that has more than a susceptible host: pigs and wild boars in Europe, with the situation in Africa being much more complicated, because there are two natural reservoirs: the Ornithodoros ticks and warthogs.
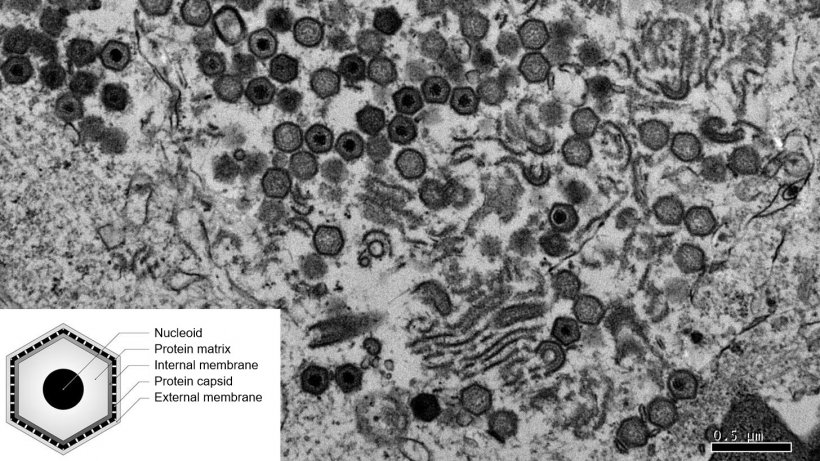
Nevertheless, and this is a completely personal assessment, what has made it even more complex is our historic inability to research this subject in a solid and coordinated way, with the closure of the best laboratories at the time (early 2000), because it was deemed that the virus was no longer a threat. And now we must rush.
At what stage is the current research for the development of a vaccine against ASF? With which guidelines are you working? When will it be possible to have a vaccine against the ASF virus?
Regardless of what has been recently described, there have been great steps forward in the research of vaccines against ASF, and we are nearer than ever with regard to giving good news. With respect to the kind of vaccine we could say, in broad terms:
- The classic inactivated vaccines do not work against the ASF virus (ASFV), at least in the way that they have been formulated so far, very probably due to their inability to produce cytotoxic T-cells, that are necessary to eliminate the infected cells.
- The live attenuated vaccines, whether with classic methods or through genetic manipulation, they provides fantastic protection levels, at least against the parental virus. As of today, this strategy is the one that is considered to be nearer to the market, and almost all the research groups regarding this matter have put their hopes in it, as shown in a report of the European Commission recently drafted by experts in the subject. It will not be easy, because the current prototypes, including the ones developed in the USDA's laboratories in Plum Island and the ones obtained through the cooperation between the Severo Ochoa Molecular Biology Centre and the CReSA-IRTA will have to be improved in the immediate future, essentially from the standpoint of their safety and their DIVA capability (that allows to distinguish between vaccinated and naturally infected animals). The consortium made up by the USDA-ARS and CReSA-IRTA guarantees the optimisation of the prototypes. The interest sparked in the business sector and the funding opened by the EU allow for optimism regarding the possibility of having an attenuated vaccine in the market within 5-10 years, always considering that these deadlines are offered by an optimistic scientist with little knowledge on corporate marketing.
- Attenuated vs subunit vaccines. The same report of the European Commission encourages us to carry on working in the vaccines of the future based on subunits. The complexity of the virus, that has more than 200 different proteins, and the difficulty of administering them in vivo so they are presented optimally to the immune system, recommends being more conservative regarding the commercialisation periods. Despite their enormous advantages regarding safety and DIVA capability, we must consider the low efficacy shown up to now against a lethal ASFV strain. We must carry on working alongside, identifying both the antigens that protect the virus and the immunological mechanisms responsible for protection. In summary, more investment is needed for basic research.
With the information available so far, which would be the expectable level of efficacy of the vaccine?
It is always difficult to extrapolate the laboratory results to the field. The only thing that we can say today is that the vaccine prototypes available in our laboratories protect 100% of the pigs against an EXPERIMENTAL CHALLENGE with Georgia, understanding protection as the ability to avoid death (the control animals die within 10 days). It is true that some animals show, for a brief period, a reduced amount of virus detected in blood and nasal secretions when compared to controls.
Which targets must this vaccine fulfil?
A crucial matter is the safety level and the DIVA capability. We must not ignore that it is a live vaccine, so it must be used with great safety guarantees. The use of vaccines based on subunitis would be much easier and less restrictive.
Although the injectable administration will very probably be better accepted by the industry, we should not forget a formulation for its oral administration, as a bait, to wild boar, in a similar way to the vaccines against tuberculosis or classical swine fever that are being tested.
Considering the impact of the disease in the last years, are there enough global resources set aside for research?
Maybe more attention is being paid nowadays to this matter and more money is being allocated for actions, but this means nothing if it is not sustained over time.
The criticism is not only to the authorities, that frequently have not taken this disease seriously enough, but also researchers, because we sometimes consider these opportunities as fishing grounds for funds without implying ourselves to the fullest in these difficulties. The limited world resources, in general, should make us reconsider how to use them rationally. I take advantage, from here, to thank publicly the funds invested by the Spanish government since 2004 in our line of research, believing in it even before than the ASF virus directly threatened our economy again. This has allowed companies like Boehringer Ingelheim (initially) and now others to become interested in our progress. We feel very lucky.












