Although Post-Cervical Artificial Insemination (PCAI) is a recently introduced technique, its discovery dates back to 1959, when the use of this methodology in pigs was described for the first time by J.L. Hancock. This technique was forgotten until only a few years ago, when its implementation really takes off at farm level. Although its use is widespread worldwide and it is a relatively easy to implement reproductive technology, we must not fail to consider some critical points in order to maximize efficiency. First of all, we should note that, at the present time, the technique must be applied to those sows that have farrowed at least once. Heat status in these females must be optimal (Figure 1A), and to make sure it is, we must consider the reproductive physiology of the female: maximum dilation of the cervix occurs during the oestrus phase close to ovulation, at which time the inner cannula can pass through the cervix without difficulty and reach the corpus uteri.
On the other hand, the spotter boar will be present during the diagnosis of oestrus (Figure 1A), but not during insemination, thus reducing cervix contractions, which facilitates the introduction of the inner cannula. A waiting period between heat detection and insemination is recommended.

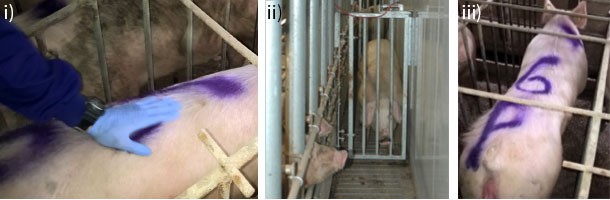
Figure 1. A) Heat: i) Identification of the sows that are in heat is done by observing their reaction to the pressure exerted by the farmer on their backs, and (ii) the presence of the spotter boar. iii) Presence of various signs of heat in the sow —such as standing reflex, pricked ears, and an oedematous and reddened vulva— can be noticed.
Hygiene is another important factor to consider during PCAI (Figure 1B.) Given that the boar’s penis and the sow’s uterus are designed to facilitate deposition of semen in the cervix, which acts as a barrier to prevent the passage of microorganisms into the uterus, the insemination location used in PCAI is "non-physiological". Therefore, it is advisable to thoroughly clean the vulva to avoid dragging particles and microorganisms into the female genital tract during insertion of the insemination device.
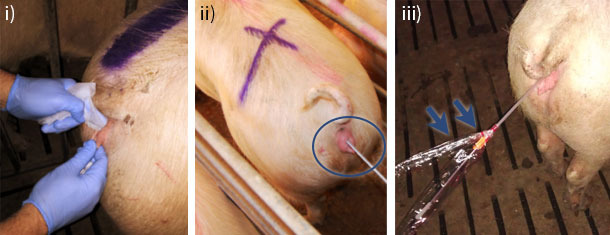
Figure 1. B) Hygiene: i-ii) The vulva must be clean prior to insemination, so the use of wipes is recommended, cleaning always from the inside to the outside of the vulva. iii) Retaining the catheter packaging can prevent soiling of the insemination device, thus increasing hygiene.
Once the female is ready, we will wait 1-2 min for the sow's cervix to relax, and then proceed with the insertion of the cervical inner cannula without difficulty. When inserting the cannula inside the female, we must ensure the insemination equipment is positioned properly (Figure 2A.) To do this, we push the catheter-cannula assembly cranially. If the cannula does not retreat in relation to the catheter, positioning is correct.
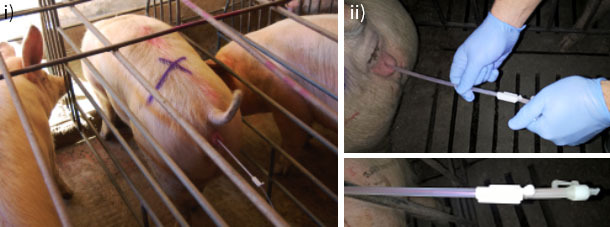
Figure 2. A) Insemination device: i) Following the introduction of the first cervical catheter, a period of time must be allowed for the cervix to relax. ii) After a few minutes, the inner cannula can be inserted completely and without difficulty. Once the catheter / cannula device is correctly positioned, introduction of semen must be carried out without delay, so as to allow proper distribution within the sow's uterus.

Figure 2. B) Reflux: i-ii) Reflux during PCAI should be nil or nearly nil; however, the release of semen through the vulva is common after insemination. This reflux can be collected, for further analysis, using bags attached around the vulva.


Figure 2. C) End of insemination: i) The cannula is removed immediately. ii) The inner cannula is inspected after passing through the cervix to ensure it maintains its characteristic curvature (not folds).
Once the catheter-cannula is positioned properly we are ready to introduce the seminal dose, the following aspects of which need to be taken into account. Results obtained in PCAI with seminal doses with a sperm concentration between 1000 × 106 and 2000 × 106 in a 26-60 ml have been found to be similar to those obtained through traditional cervical insemination (3000 × 106 sperm cells / 80-100 ml.) The use of doses with a higher or lower sperm concentration in PCAI will depend on many other factors, that could be the subject of another article. On the other hand, it is important to consider sperm quality, because the use of fewer sperm cells means that any reduction in quality can have serious consequences on fertility and prolificacy. The insemination dose must be introduced quickly to ensure its distribution within the uterine horns. There should be no reflux during PCAI (although it is common in the first minutes post-insemination – Figure 2B.) If reflux is noticed through the vulva or catheter / cannula, it might be an indicator that the cervix is not completely relaxed, so we'll wait a few more minutes before repeating the process. Another possible cause is that insemination has been carried out too early (at the beginning of oestrus) or too late (at the end of oestrus), when the cervix is not fully open. Once the seminal dose has been deposited, the cannula needs to be withdrawn immediately in order to break the connection of the uterus with the outside environment, thus preventing the introduction of pathogens. After removing the inner cannula, it is recommended to check that its curvature correlates to the anatomical shape of the cervix, and that it has no folds (Figure 2C), indicating that insemination has been performed correctly. If we notice certain opposition to the introduction of the cannula, it is important not to force penetration because that may damage the uterus lining causing it to bleed; this blood would be seen when removing the insemination device. Still, reproductive parameters may be very similar among females in the absence or presence (not in big quantities) of blood during insemination, although of course, it is not the ideal situation.
In summary, given that small errors in any of the above factors can cause the end result of the application of this technique to differ from expectations, every single one of the hazards and critical control points of each of them needs to be analysed in an orderly manner (Table 1).
Table 1. Checklist of factors to consider during post-cervical insemination: critical control points.
| Critical points | Description | References | |||
| 1. Type of female | Multiparous and primiparous. | Watson and Behan, 2002 Diehl et al. 2006 Hernández-Caravaca et al. 2012 Sbardella et al. 2014 |
|||
| 2. Heat | Optimal heat status – Maximum dilation of the cervix | Rigby, 1967 | |||
| 3. Spotter boar | Heat detection and uterine stimulation. Absent during post-cervical insemination. |
Langendijk et al. 2005 | |||
| 4. Hygiene | Throrough cleaning of the vulva. | Araujo et al. 2009 García-Vázquez et al. 2015 Hernández-Caravaca et al. 2015 |
|||
| 5. Device | Proper positioning of the catheter / cannula. | Watson and Behan, 2002 | |||
| 6. Seminal dose | Concentration and volume. Sperm quality. | García-Vázquez et al. 2015 Hernández-Caravaca et al. 2012 y 2015 |
|||
| 7. Reflux | Absent (or scant) during post-cervical insemination. Frequent after insemination. | Steverink et al. 1998 Dallanora et al. 2004 Mezalira et al. 2005 Hernández-Caravaca et al. 2012 |
|||
| 8. Blood | Do not force the insertion of the inner cannula to avoid injury. | Serret et al. 2005 Sbardella et al. 2014 |
|||











