Lameness is a highly prevalent production disease. It is responsible for 7 to 15% of sows culled in different countries (Gill, 2007). However, farm surveys including moderately lame animals show the number of lame sows is far greater. A recent study found that 39% of replacement gilts, 41% of pregnant gilts and 42% of multiparous sows were lame (Quinn, 2013). In addition to the obvious welfare consequences lameness can be detrimental to farm profitability. In a previous article we detailed the main causes of lameness in sows. In this article however we examine the nutritional effects relating to sow lameness in more detail as nutrition and limb health are closely linked, particularly in relation to composition of the diet and nutritional strategies.

Diet Composition
Vitamins & mineral supply
Supplying the correct vitamins and minerals in the right quantities is crucial to limb health in sows (van Riet et al., 2013). Keratinisation of the hoof epidermal cells to make horn requires a suitable supply of minerals, vitamins and trace elements. If the supply of nutrients is inadequate hoof horn of reduced quality is produced, resulting in the hoof being more vulnerable to physical, chemical and microbial damage from the surrounding environment. Trace elements; zinc, manganese and copper are central in hoof health, enzyme system and immune system functioning. The addition of an organic chelated trace mineral complex of zinc, copper and manganese to the diet of breeding sows reduces heel erosion, heel overgrowth and white line lesions after supplementation (Anil,2011; Bradley, 2010). This suggests that such minerals may help to reduce the prevalence of lameness in sows (van Riet et al., 2013). Calcium and phosphorus are necessary for the development and maintenance of bone. During gestation and lactation the calcium requirements increase and calcium reserves in the bones may temporarily be used to meet this demand, if a long term deficiency results, long term bone strength is reduced making sows more vulnerable to bone fracture. Gilt, gestation and lactation diets therefore need to supply adequate calcium and phosphorous levels. Biotin is one of the most widely investigated vitamins in relation to claw hardness, playing an important role in the formation of keratinised tissues which include the skin, claw horn and foot pads, especially in young sows. Additionally the main nutritional causes of osteochrondrosis in pigs is a result of vitamin deficiency (Vitamin C, A, D and Biotin), copper deficiency and excess zinc in the diet (van Riet et al., 2013).
Fibre Content
Loose housing is associated with higher levels of lameness than stalled systems due to the inherent need to establish a dominance hierarchy, thus an increase in fighting behaviour, and therefore increasing lameness (Quinn , 2014). Gestation diets are restrictively fed to sows to prevent excess fat levels, however this can result in inadequate satiety and high levels of feeding motivation thus increased levels of frustration. Diets high in fibre (i.e. 8% +) promote a feelings of satiety as a result of better ‘gut fill’ enabling the sow to feel fuller for longer, thus satisfying her inherent need to forage which in turn reduces frustration, restlessness and aggression (Stewart et al., 2010, 2011). The precise fibre requirements in the diet will vary according to the frequency of feeding and the source of fibre being used.

Figure. 1 Various types of loose sow housing. In all cases the increase in fighting increases lameness.
Gilt Feeding Regime
When considering nutritional influences on sow lameness you need to take a step back and look at gilt development. Young replacement animals are particularly susceptible to lameness. Between 15 and 20% of gilts and first and second parity sows are culled due to lameness (Lucia et al., 2000). This represents a major economic concern as a sow does not become cost neutral until after she has had her third litter. An Irish study found that 39% of replacement gilts were lame on entry to the breeding herd (Quinn et al., 2013). A gilt who has compromised limb health on entry to the breeding herd is more vulnerable to early removal as a result of lameness and more vulnerable to the negative production affects associated with it.
The effect of gilt nutrition on future reproductive performance has been widely investigated but the potential effects of gilt feeding strategies on limb health has been examined to a much lesser extent. Gilt nutrition during the development phase is often inadequate. Feeding diets formulated for finisher pigs until service or diets formulated for pregnant sows to gilts from selection at 100 kg is nutritionally inadequate for their development. A gestating sow diet is formulated for a sow that has finished growing and a finisher diet is formulated for fast growth rates, high lean meat deposition and cost efficiency. High growth rates are linked with several pig welfare issues, and so are counter-productive for growing female breeding animals. In contrast diets specifically formulated for gilts include the nutrient requirements for bone development and fat deposition. Replacement gilts require higher levels of calcium and phosphorous for bone mineralisation to prevent bone weaknesses and as a backup source of Ca and P for the litter if needed during gestation and lactation. Studies have found that feeding a diet specifically formulated for developing gilts (14.04 MJ of DE/kg, 0.75% lysine) from ~65-70 kg to 2 weeks before target service at ~140 kg, reduces lameness levels when both restrictively (2.25kg) and ad-libitum fed, compared to feeding either finisher or gestation diets (Quinn et al., 2015). Restrictively feeding a gilt developer diet also resulted in a reduced prevalence of osteochrondrosis, as shown in the figure, and claw unevenness, while no such joint lesion and claw benefits were observed when ad-libitum feeding during development, however an increase in areal bone mineral density was observed (Quinn et al., 2015).

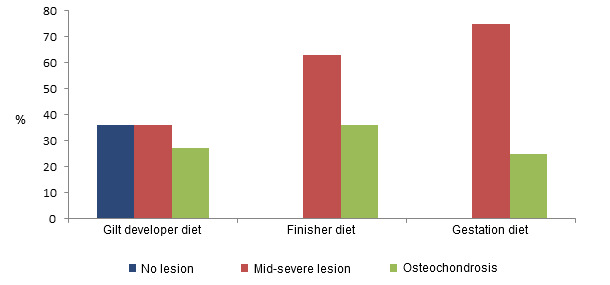
Figure 2. Prevalence of claw lesions in gilts depending on the diet fed during their development. Feeding gilts a developer diet results in a reduced prevalence of lesions Quinn et al., 2015.





