History
Ear necrosis is a fairly common yet enigmatic malady encountered in swine production globally. Presumed causes of ear necrosis are many and generally speculative; indeed, there are many risk factors associated with ear necrosis including trauma from pen mates or the environment, overcrowding, commingling, high humidity, limited feeder or water space, misformulated rations, mycotoxins, viral diseases, or bacteria (Park, Friendship et al. 2013; Svartström, Karlsson et al. 2013; Weissenbacher-Lang, Voglmayr et al. 2012, Richardson, Morter et al. 1984, Karlsson, Klitgaard et al. 2014). The genuine mechanisms and pathogenesis of ear necrosis have yet to be elucidated, likely because the condition is not due to a single entity or mechanism. Analogous, if not indistinguishable presentation grossly, occurs in canines and is usually associated with vasculopathies, particularly immune-mediated vasculitis associated with infections, vaccination, and drug eruptions (Scott, Miller et al. 2001).

Ear necrosis is characterized by bilateral or, less likely, unilateral necrosis of the tip and posterior edge of the pinna. Lesions range from mild, superficial erosion to more severe inflammation with exudation, ulceration, and necrosis (Figure 1). The subsequent compromise to the epidermis allows access to dermal vessels leading to the systemic spread of secondary pyogeneic bacteria resulting in arthritis, embolic pneumonia, abscesses and condemnations at slaughter.
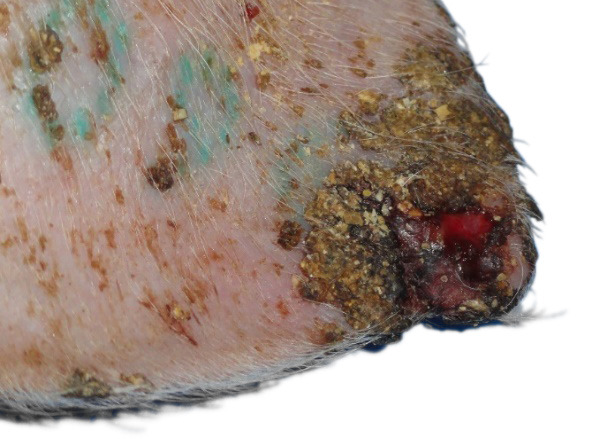
Figure 1 – Ulceration, crust formation and thickening of the epidermis.
Cases presented to the Iowa State University Veterinary Diagnostic Laboratory suggest that a majority of herds in the United States have had sporadic cases which occur unpredictably and with large variations in frequency of outbreaks, morbidity, severity of lesions and overall impact on the population.
A priori hypotheses
- Hypothesis #1: The systemic spread of one or more etiologic agent(s) is associated with ear necrosis.
- Hypothesis #2: There is immune complex deposition in small caliber vessels leading to vascular compromise and ear necrosis.
- Hypothesis #3: There is an idiopathic neuropathy/vasculopathy causing ear necrosis. The levels of vasoconstrictors and/or vasodilators is different between affected and non-affected pigs.
- Hypothesis #4: There is a primary bacterial etiology that colonizes the epidermis leading to ear necrosis.
Recent results
The objective of recent ear necrosis investigations conducted at farms located in the Midwest region of the United States was to characterize the progression of lesions and detect the presence of potential pathogens. Samples were obtained for histopathology in all cases and molecular diagnostics and metagenomics in a single investigation.
Histopathology of the epidermis and dermis of acutely affected pigs revealed deep dermal perivascular inflammation with minimal changes to the epidermis (Figure 2). Changes to the epidermis included acanthosis and orthokeratotic hyperkeratosis. In one investigation, eosinophils, vasculitis, thrombosis, and epidermal necrosis predominated. Changes in chronically affected pigs included ulceration, irregular acanthosis, granulation tissue formation, and deep dermal thrombosis (Figure 3). Chronic changes are similar to those described earlier (Jensen 2009, Richardson, Morter et al. 1984)
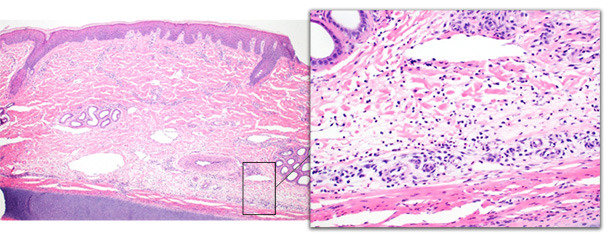
Figure 2 – Ear tip biopsy from an acutely affected pig. The deep dermis is expanded by edema and moderate numbers of neutrophils with minimal epidermal changes.

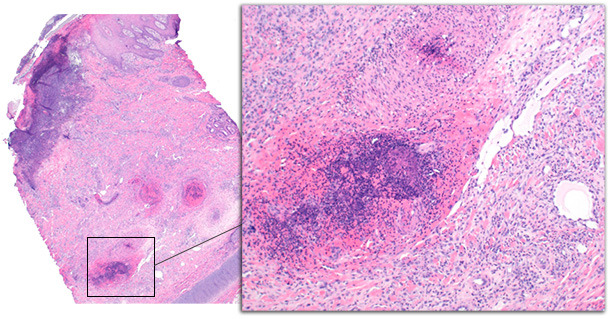
Figure 3 – Ear tip biopsy from a chronically affected pig. Epidermal changes include ulceration with subjacent inflammation, serocellular crust formation, and acanthosis of the epidermis. The deep dermis is expanded by granulation tissue and hemorrhage. Deep dermal vessels are disrupted by thrombosis and vasculitis.
Polymerase chain reaction (PCR) results for the detection of Staphylococcus hyicus exfoliative toxin genes were negative; although the PCR was optimized for extracts from culture rather than extracts generated from tissue samples (Onuma, Uoya et al. 2011). Metagenomic analysis of the epidermis and dermis using amplification of the V3-V4 hypervariable region of the bacterial 16S rRNA gene revealed statistically significant differences between pigs with chronic ear necrosis and pigs without ear necrosis in alpha (richness) diversity (P = 0.003); this means that the number of species identified based on the number of sequences sampled for each of the groups was less in the chronic group. Specifically, the relative abundance of Actinobacillus spp., Fusobacterium spp., Streptococcus spp., and Staphylococcus spp. was statistically significantly different between affected (acute and chronic groups combined) and nonaffected groups (Figure 6).
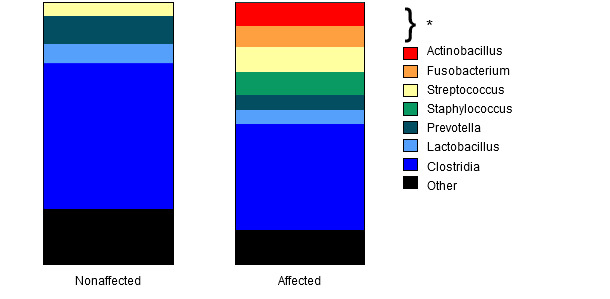
Figure 6 - The relative abundance by group of bacterial genera and the class Clostridia.
A statistical analysis to assess the biologic importance of each significant taxa was used. When comparing pigs with ear necrosis (acute and chronic groups combined) versus pigs without ear necrosis, 209 taxa where determined to be biologically significant. When comparing between acute, chronic, and nonaffected ears, 24 taxa were determined to be biologically significant. Within this group, two potentially septicemic bacteria were identified as biologically significant in samples from acutely affected pigs: Actinobacillus spp. and Haemophilus spp.
Generally in these case investigations, the presence of dermal inflammation with minimal epidermal changes is suggestive of an “inside-out” rather than “outside-in” process and could suggest compromise of the vascular supply leading to colonization by and lesion progression due to opportunistic bacteria. Metagenomic analysis has revealed that bacterial communities change significantly as lesions progress and the role of the Actinobacillus and Haemophilus genera as primary etiologies or secondary invaders due to vascular compromise requires further investigation.
Future directions
A prospective study may help elucidate possible mechanisms involved in the development of ear necrosis. Multiple assays are likely needed including the tried-and-true techniques of immunohistochemistry and electron microscopy for the detection of immune complexes, culture for the characterization of bacterial toxins by PCR, as wells as the more novel techniques of metagenomics and transcriptomics to further characterize the ear microbiome of affected and nonaffected pigs and relative abundance of vasodilators and vasoconstrictors, respectively. As always, sample selection and stage of disease are imperative. Chronic lesions provide minimal information concerning the potential inciting etiologies.
Summary
Ear necrosis remains an enigma. The use of different techniques in an orchestrated manner may elucidate mechanisms and/or etiologies allowing for the implementation of preventative measures and targeted treatments.


