On July 27th, 2016, the EMA (European Medicines Agency) published the document on the use of colistin in the European Union, establishing a maximum use of 5 mg/Population Correction Unit (PCU) that the member states will have to reach in a period of three years, the ideal figure being 1 mg/PCU.
After this decision taken by the EMA, representatives of the national associations of veterinarians and professionals in the pig production sector of Spain met on September 2016 with the aim of reaching an agreement to reduce the consumption of colistin. As a result of this meeting, the “Agreement for the Voluntary Reduction in the Consumption of Colistin in the Pig Production Sector in Spain” was drafted.

Up to now, in Spain 44 companies (representing some 70% of the national pig production) have adhered to the voluntary program for the reduction of colistin.
Although the program’s aim is to reduce the consumption of colistin in pig production, it also expects to control the consumption of alternative antibiotics, avoiding the increase in the consumption of neomycin and/or apramycin as possible alternatives to colistin.
Results
| 2015 | 2016 | 2017* | |
| mg/PCU Colistin | 51.09 | 23.91 | 9.00 |
| mg/PCU Neomycin | 38.83 | 25.51 | 14.81 |
| mg/PCU Apramycin | 1.04 | 1.37 | 1.83 |
* data in the first semester of 2017
According to these data, the consumption of colistin between 2015 and the first semester of 2017 has dropped by 82.37%, whilst the consumption of neomycin (a possible alternative to the use of colistin) has also shown an important decrease. The consumption of apramycin, also a possible alternative to the use of colistin, has remained relatively stable, with a small increase by 0.5 mg/PCU, that is not alarming, but that shows that its consumption must be monitored.

Evolution of the consumption of colistin in mg/PCU.
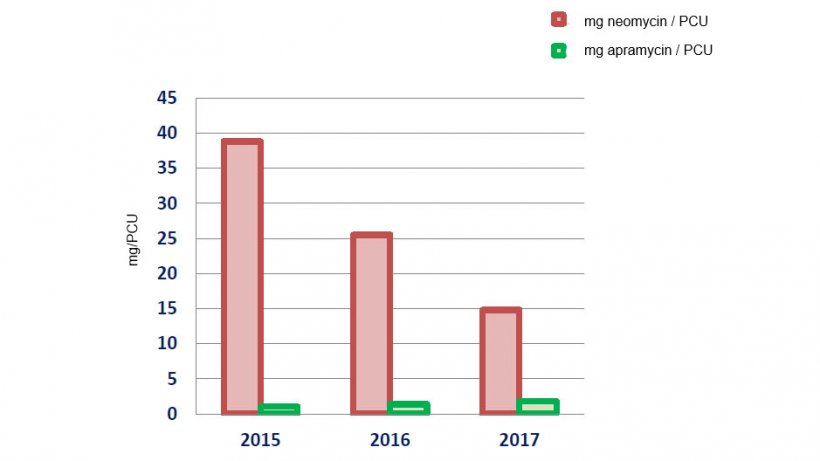
Evolution of the consumption of neomycin and apramycin in mg/PCU.
First Report Program Reduce Colistin - AEMPS/ Spain.





