Porcine Circovirus Type 2 (PCV2) is the causative agent of Porcine Circovirus Disease (PCVD). In North America, prior to the introduction of commercial vaccines, PCVD caused significant economic losses to producers from increased mortality, culling rates, and poor growth in growing pigs. The effects of PCV2 in breeding herds have not been consistently identified. There have been numerous field reports of PCV2 related reproductive failure beginning as early as 1999. In general, these field reports suggest increased rates of mummified and stillborn fetuses and in some cases decreased conception rates. In every case, no impact on economics was reported, but these cases clearly demonstrate that PCV2 can cause of reproductive failure.
The causes of reproductive failure appear to be due primarily to fetal infection with the virus. When during gestation the fetuses are infected has a significant impact on the clinical signs observed. Sows that are infected in the first trimester of pregnancy or immediately prior to breeding as gilts will produce embryonic death and subsequent return to estrus or low number of pigs born alive. Infection later in pregnancy tends to produce mummies and stillborns.

The impact of PCV2 on breeding herds may not be as dramatic as other diseases such as Porcine Reproductive Respiratory Syndrome (PRRS), but the evidence demonstrates that PCV2 can produce economic losses in breeding herds. As an example we will explore what happened in one large North American breeding herd after PCV2 was introduced.
Abruptly, mummified fetuses increased from approximately 2 percent to over 10 percent with no change in stillborn pigs or abortions. Sows farrowed at the expected time (114 to 115 days of gestation) but would have variable numbers of mummified and live born pigs (Table 1). The striking part was that in spite of high numbers of mummified pigs in a litter, the pigs that were born alive were normal and survived to weaning at a normal rate. At the same time there was a corresponding decrease in the total number of pigs born per litter that was a result of both increased fetal morality and a reduction in accuracy in counting pigs at birth due to many small mummies (less than 2 cm) that were deposited in the fetal membranes at the end of farrowing which were not observed by caretakers. Interestingly, there was an increase in the number of irregular returns (any return that occurs between 24 and 37 days after breeding and suggests pregnancy failure) starting 22 weeks before the increase in mummies suggesting that the initial infection was months before the elevated mummies were seen. There were no other changes in performance metrics (farrowing rate, stillborn percent, sow mortality) over this time period. Following the 10-week spike in mummies, the mummy rate returned to a rate that was twice the rate prior to the outbreak.
Table 1: Clinical History of a swine breeding herd farrowing only primiparous animals that experienced a ten week
outbreak of PCV2 related reproductive failure characterized by an increase in mummified fetuses.
The listed interventions during development and breeding are for each listed farrowing cohort.
| Farrowing week | Pct mummies of total- born pigs | Interventions during development and gestation of gilts that farrowed during week | Week of gestation when vaccinated PCV2 vaccine 1 (Mass vaccine in 2008-48) | |||
| Date | Week numbers | PCV2 vaccine at weaning (3 weeks of age) | PCV2 vaccine pre-breeding | Acclimation period prior to entry into gestation barn | ||
| 5/1/08 | 18-20 | 2.5% | None | Full Dose Vaccine 2 | 4.5 weeks | n/a |
| 5/22/08 | 21-23 | 2.5% | None | 1.5 weeks | ||
| 6/12/08 | 24-28 | 2.5% | 1/2 Dose Vaccine 1 | |||
| 7/17/08 | 29-36 | 2.5% | Full Dose Vaccine 2 | |||
| 9/11/08 | 37-45 | 9.5% | ||||
| 11/13/08 | 46 | 9.5% | Post Farrowing | |||
| 11/20/08 | 47 | 9.5% | Post Farrowing | |||
| 11/27/08 | 48 | 9.5% | Post Farrowing | |||
| 12/4/08 | 49 | 5% | Post Farrowing | |||
| 12/11/08 | 50 | 5% | Post Farrowing | |||
| 12/18/08 | 51 | 5% | 13 | |||
| 12/25/08 | 52 | 5% | 12 | |||
| 1/1/09 | 1 | 5% | 11 | |||
| 1/8/09 | 2 | 5% | 10 | |||
| 1/15/09 | 3 | 5% | 9 | |||
| 1/22/09 | 4 | 5% | 8 | |||
| 1/29/09 | 5 | 5% | 7 | |||
| 2/5/09 | 6 | 5% | 6 | |||
| 2/12/09 | 7 | 5% | 5 | |||
| 2/19/09 | 8 | 5% | 5 | |||
| 2/26/09 | 9 | 5% | 5 | |||
| 3/5/09 | 10 | 5% | 5 | |||
| 3/12/09 | 11 | 5% | 5 | |||
| 3/19/09 | 12 | 5% | n/a | |||
With the increase in mummies a series of diagnostics were conducted. A summary of the diagnostic results is summarized in Table 2. In the initial diagnostic submission all of the fetuses had distended abdomens that were suggestive of heart failure. To better understand the cause of the apparent heart failure, additional cases were submitted and immunohistochemistry (IHC) for PCV2 on both fetal tissues and the placenta was conducted. IHC demonstrated very high amounts of PCV2 in myocardial (Figure 1) and splenic tissue.
Table 2: Summary of diagnostic results for swine breeding herd that only farrowed
primiparous animals during an outbreak of increased mummies.
| Pig age | Late term fetus | Late term fetus | Pigs at farrowing |
| Date | 11/6/2008 | 11/18/2008 | 12/13/2008 |
| Samples | Mummy | Mummies from 5 litters | Stillborn and live pigs from 5 litters |
| PCV2 PCR | Pos | Pos | Pos |
| PCV IHC | Pos | Pos | |
| Fetal antibodies | Pos | ||
| Parvovirus | Neg | Neg | Neg |
| Leptospira | Neg | Neg | |
| PRRSV | Neg | Neg | |
| PCV Type | Not determined | NA-PCV2a | NA-PCV2a |
| PCV2 RFLP Pattern predicted from sequence | Not determined | 3-2-1 | 4-2-2 |
| Histogical lesions | None | None | Myocardial degeneration, necrosis, and fibrosis, multifocal, marked with mild lymphocytic myocarditis |
| Comments | Significance of PCV finding is not known. The presence of fetus with distended abdomens that is suggestive of heart failure that may be congenital. | ICH was +2 to +4 pos on tissues but placenta was negative. | Comments: Microscopic lesions are compatible with in utero PCV2 infection. |
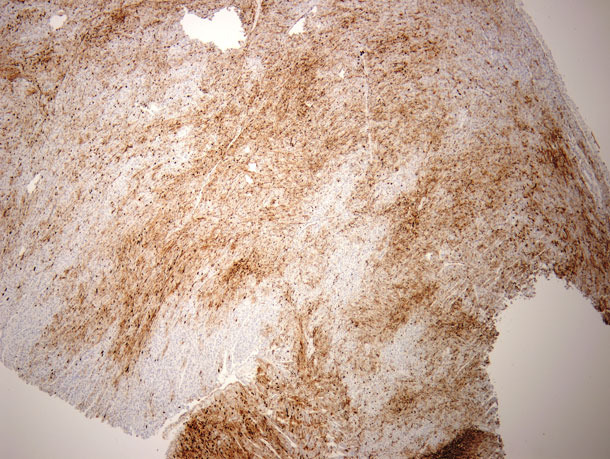
Figure 1: Immuno-histo-chemistry (IHC) staining of heart muscle tissues for PCV2 from a 1 day old pig that was born alive in a litter that consisted of 8 mummies and 4 live born pigs. The dark brown areas represent positive staining against PCV2, the ligth brown areas are normal heart muscle.
Numerous changes we made in PCV2 management strategies prior to the outbreak. These are summarized in Table 1. In response to the outbreak mass vaccination of the entire herd was completed 8 weeks after the outbreak started. It appears, based upon analysis of SPC charts, that vaccination was not successful in reducing the rate of mummies as no SPC signals were generated from the mass vacation of the herd. Vaccination of gilts prior to breeding was restarted in early 2009 and 20 weeks later (when the vaccinated gilt started to farrow) the mummified fetus rate remained >4.5% and remained at that level until the acclimation period was extended to a minimum of 4 weeks. The herd had maintained pre-breeding PCV2 vaccination as a preventative.
When gilts are introduced into the breeding herd without prior exposure, PCV2 can play a significant role in reproductive disease. Under these conditions, routine control measures such as vaccination do not appear to be effective in eliminating clinical signs within the breeding herd. Altering animal flow to insure exposure to PCV2 prior to breeding, as part of an acclimation program, is necessary to minimize the risk of PCV2 associated reproductive disease.