Methods to detect antibodies for PRRSV
The indirect fluorescent antibody (IFA) test, serum virus neutralization (SVN) test, immunoperoxidase monolayer assay (IPMA), and enzyme-linked immunosorbent assay (ELISA) have all been used for the detection of antibodies specific for PRRSV. The IFA, SVN, and ELISA are currently available in most North American veterinary diagnostic laboratories, while IPMA has been extensively used in Europe. All tests are considered to be specific with varying degree of sensitivity. Typically IFA, IPMA and ELISA are for detection of total antibody of IgG isotype specific for PRRSV while SVN test is used to detect functional antibody related to immunity or immune protection. IFA test and ELISA have been adapted to detecting other isotypes such as IgM or IgA (Figure 1). PRRSV-specific IgG antibody which can be detected by IFA, IPMA or ELISA is produced in infected pigs between 7 and 14 days post-infection; however, SVN antibody is detectable as late as 1 to 2 months after infection (Figure 2). Generally PRRSV antibody from active infection can be reliably detected for 3 to 4 months after infection and can last 6 months up to 1 year post outbreak.

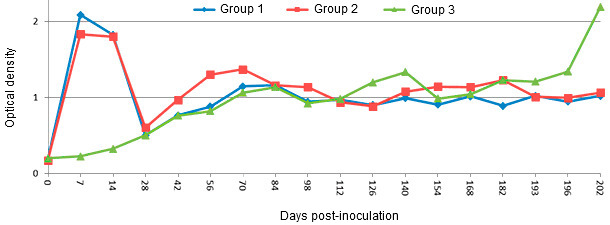
Figure 1. IgM response in young pigs after experimental PRRSV infection as measured by IDEXX PRRS ELISA 2XR with modifications. Group 1 and 2 were inoculated with PRRSV type 2 intranasally on day 0. Group 1 was re-inoculated on 196 days post inoculation (dpi) whereas group 2 was not. Group 3 was not inoculated with PRRSV until 196 dpi. PRRSV specific IgM antibodies are detectable by 7 dpi and quickly decline after 14 dpi. (Nezami S and Yoon KJ, unpublished data)
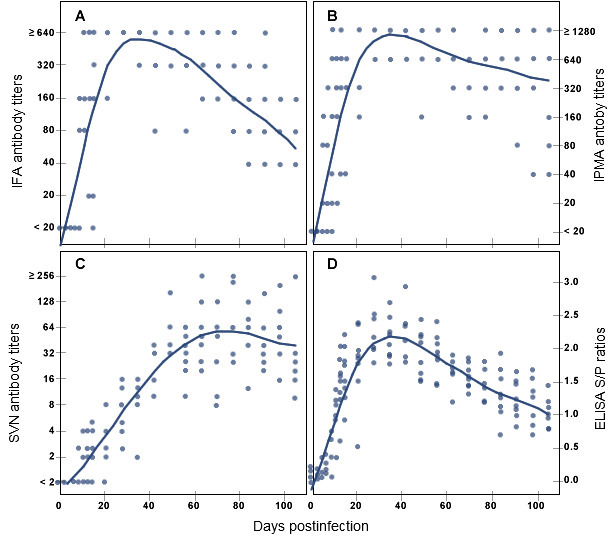
Figure 2. PRRSV specific antibody response in pigs over time after experimental infection as measured by indirect fluorescent antibody assay (A), immunoperoxidase monolayer assay (B), serum-virus neutralization assay (C) and enzyme-linked immunosorbent assay (D). (Adopted from publication by Yoon K-J, et al.: Characterization of the humoral immune response to PRRSV infection. Journal of Veterinary Diagnostic Investigation, 7:305-312, 1995)
IFA and IPMA tests utilize virus-infected cells as antigen (Figure 3). An advantage of the IFA test and IPMA compared to ELISA is that the magnitude of the antibody titer can be determined. However, the endpoints of IFA antibody titers often vary among technicians and laboratories since interpretation is subjective. Furthermore, test results or endpoint antibody titers will vary depending on the degree to which the PRRSV strain used in the assay differs antigenically from the isolate that infected the pig.
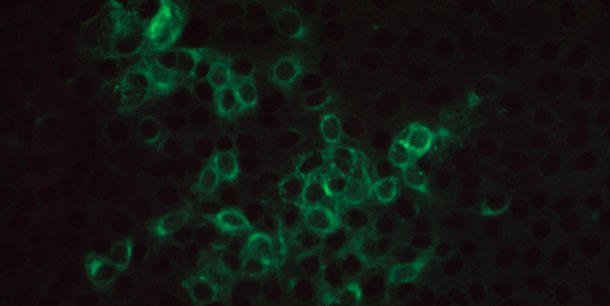
Figure 3. Photomicroscopy of positive immunofluorescence on PRRSV-infected MARC145 monolayer incubated with a pig serum containing PRRSV specific IgG antibody and stained with goat anti-porcine IgG antibody conjugated with FITC.
The ELISA is believed to be more sensitive than IFA test and IPMA. Several ELISA formats have been described: an indirect ELISA using a sample to positive (S/P) ratio system, an indirect ELISA using direct OD values, and a blocking ELISA. At present there are several commercially available ELISA kits for detection of serum antibodies specific for PRRSV (Figure 4). Uniformity in the manufacturing of the kit and a high degree of automation in performing the test in the diagnostic laboratory result in less variation in the results from the commercial ELISA compared to other assays. Other advantages of the commercial ELISA may include: a) detection of antibody against both types of PRRSV; b) fast turnaround time; and c) licensure. Lately ELISA platform has also been adapted to testing oral fluid samples for PRRSV antibodies as part of herd monitoring.

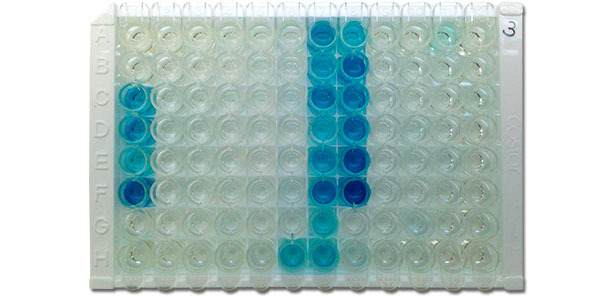
Figure 4. Photography of IDEXX ELISA X3 plate showing positive (blue color) and negative (no color) results. Color density correlates with antibody level of serum, i.e., the darker color is the higher antibody titer is. (Photo courtesy: Dr. David Baum, Iowa State University Veterinary Diagnostic Laboratory)
The SVN test is also considered a specific test, but previous studies have suggested that SVN is less sensitive than IFA and ELISA. The low sensitivity of the test is primarily due to the fact that neutralizing antibodies against PRRSV develop late and slowly. The SVN test is best considered a research tool rather than a routine diagnostic test because of its laborious nature. As with IFA and IPMA, test results are greatly influenced by the degree of antigenic relatedness between the isolate employed in the test and the isolate infecting pigs.
Interpretation of serological results in PRRS diagnosis
Serology can be helpful in confirming the presence (i.e., seropositivity) and status (e.g., high levels of antibody in recent infection) of PRRSV infection in the herd. Serological information from a single sample is not sufficient for diagnosing clinical PRRS in an individual animal because PRRSV infection is highly prevalent in swine herds. The possible presence of maternal antibody should be considered when interpreting serology results on young pigs. It is also important to bear in mind that the current serologic assays used in the diagnostic setting cannot routinely differentiate vaccine-derived antibodies from field isolate-derived antibodies. Furthermore, the occurrence of suspected false PRRSV positive serology has been documented even with commercial ELISA kit. Negative PRRS serology on samples at one point in time has several possible interpretations as well: a) The pigs were not infected with virus; b) The pigs were recently infected with virus and have not yet seroconverted; c) The pigs were infected with virus, but have become seronegative; and d) The result was negative because of the low sensitivity of the test or laboratory error. Therefore, if using single point samples, PRRS serology must be used in conjunction with valid population sampling methods to determine if a herd has been exposed to PRRSV, not whether individual animals are infected.
It is generally recommended that young pigs, rather than breeding stock, be tested to determine a herd's PRRSV infection status. In single-site, farrow-to-finish swine herds, the seroprevalence of PRRSV infection is usually considered to be highest in the grow-finish unit. Serum from 10 finishing pigs is often sufficient to determine whether the herd has been exposed to PRRSV. For multi-site production systems, each stage of production represents a single population, so each site should be sampled and a series of blood samples from various stages of production is useful to determine the age that PRRSV infection is most common.
Serodiagnosis of PRRSV infection as the cause of reproductive failure or respiratory disease may be achieved by showing seroconversion using paired serum samples or a change in antibody titer (i.e., rising antibody titer) in paired serum samples. However, sows may be exposed to virus 2 or more weeks prior to the onset of clinical signs, in which case rising antibody titers may not be observed. The presence of PRRSV antibodies in fetal fluids or in blood collected from stillborn and weak piglets before ingestion of colostrum is also indicative of PRRSV infection. Nevertheless, a definitive diagnostic evaluation of PRRS with respect to clinical disease requires that serological information be interpreted in combination with results from other virus assays.








