1. Introduction
Since its discovery in 1991, porcine reproductive and respiratory syndrome virus (PRRSV) continues to cause major economic losses to the global pig industry. PRRSV infection of sows and gilts may vary from subclinical to severe reproductive failure and death of the dam. Increased numbers of abortions, weak born pigs, mummified fetuses, and small litter sizes are common. PRRS in growing pigs may also be subclinical or severe and is most typically manifest as respiratory disease and decreased weight gain. In growing pigs, PRRSV mainly targets the respiratory tract and immune systems. The virus replicates in macrophages in the lungs and in several cells in the lymphoid system. Macrophages in the lungs are an important first defense in the respiratory tract. When the lungs and immune system are damaged or altered by PRRSV, the pigs become more susceptible to other respiratory and septicemic pathogens.

2. PRRSV types
The currently recognized main PRRSV genotypes are summarized in Fig. 1. In general, type 2 isolates are considered to be more virulent than type 1 isolates.
| Type 1 | Type 2 |
| Synonym: European PRRSV |
Synonym: North American PRRSV |
| Prototype: Lelystad virus |
Prototype: VR2332 Atypical PRRSV: (1998) High pathogenic (HP) PRRSV: Limited to China, (2006) |
Fig. 1. Currently recognized main PRRSV genotypes.
3. PRRSV and the porcine respiratory disease complex (PRDC)
The term porcine respiratory disease complex or PRDC is commonly used to describe the outcome of interactions of multiple respiratory pathogens (both viral and bacterial), environmental factors, type of production system and quality of management, and pig-specific factors (genetics, age, immunological status). PRRSV is considered a primary and perhaps the most important pathogen in PRDC (Fig. 2).
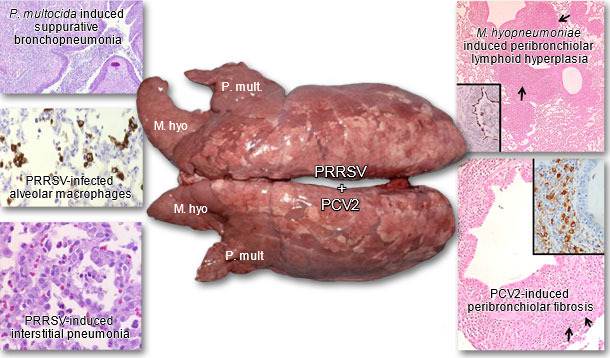
Fig. 2. Lungs from a pig suffering from PRDC. Concurrent PRRSV, PCV2, M. hyopneumoniae, and P. multocida type A infection were diagnosed. Inserts: Immunohistochemical stains for M. hyopneumoniae, PCV2 and PRRSV. Brown staining indicates presence of antigen. Images courtesy of Dr. Patrick G. Halbur.
4. PRRSV and viral co-infections of the respiratory system
Porcine respiratory coronavirus (PRCV). PRCV replicates mainly in the epithelium in the lower respiratory tract. Macroscopically, lungs may be non-collapsed and multifocally mottled-tan in color. When alone, PRCV induces mild interstitial pneumonia and mild necrotizing bronchiolitis. Under experimental conditions, PRCV and PRRSV co-infected pigs have more severe respiratory disease and lung lesions.
Swine influenza virus (SIV). This virus targets respiratory tract epithelial cells of the upper and lower respiratory tract causing damage to the epithelial lining and the mucociliary apparatus. Affected lungs often have mutlifocal dark red-to-purple consolidation. SIV also decreases macrophage function. However, when pigs were co-infected with PRRSV and SIV, SIV replication was only slightly increased by prior PRRSV infection.

Pseudorabies virus (PRV). PRV replicates in alveolar macrophages and monocytes and is often associated with necrotizing rhinitis and tracheitis. Increased disease severity and duration of respiratory disease was observed in conventional pigs concurrently infected with PRV and PRRSV.
Porcine circovirus type 2 (PCV2). PCV2 has gained importance in recent years. Prominent lymphadenopathy of tracheobronchiolar lymph nodes and congested, non-collapsed lungs are typical macroscopic findings with PCV2 infection. Microscopically, PCV2 is associated with mild interstitial pneumonia, attenuation and necrosis of airway epithelial cells, and peribronchiolar fibroplasia in lung tissues as well as lymphoid depletion. Concurrent PRRSV and PCV2 infection is a frequent scenario under field conditions and it is well known that concurrent PRRSV-PCV2 infection enhances respiratory disease and severity of lung and lymphoid lesions. In particular, PRRSV appears to enhance and prolong PCV2 infection in dually-infected pigs further complicating effective clearance of both viruses from pigs.
5. PRRSV and bacterial co-infections in the respiratory system
Streptococcus suis. This bacterium can enter the lung from the upper respiratory tract or reach the lung following septicemia. Affected lungs are often covered with fibrin. PRRSV infection increased susceptibly of pigs to Streptococcus suis under experimental conditions where researchers demonstrated PRRSV-induced damage to the pulmonary intravascular macrophages which are important for clearing the bloodstream of bacteria.
Bordetella bronchiseptica. Infection with Bordetella bronchiseptica is commonly seen in younger pigs. Grossly, well demarcated areas of dark red-to-purple consolidation with mild pleuritis can be seen particularly on the front and lower portions of the lung. Bordetella bronchiseptica also plays a role in atrophic rhinitis. In PRRSV coinfected experimental pigs, exacerbated disease was observed.
Pasteurella multocida (P. multocida). This is a common, perhaps even the most common bacterial opportunistic invader that is cleared fast from lungs under normal circumstances. Macroscopic lesions may include red-to-grey consolidation of the front and lower portion of the lung. Microscopically, P. multocida infection induces various degrees of suppurative bronchitis with often marked accumulation of exudate in airway lumens. When pigs were coinfected with PRRSV and P. multocida, no clear interaction was observed under experimental conditions. However, as PRRSV and P. multocida are among the most common swine pathogens and are frequently found together in field cases, some interaction likely occurs.
Mycoplasma hyopneumoniae (M. hyopneumoniae). Infection with M. hyopneumoniae is quite common and most pig herds are infected. Infection leads to loss of cilia, epithelial cell death, and reduced function of the mucociliary apparatus. Grossly, mild to severe, dark-red-to-tan consolidation of the lung can occur. Characteristic microscopic lesions include peribronchiolar and perivascular lymphoid hyperplasia in the lungs. Several experimental studies have shown a clear relationship between M. hyopneumoniae and PRRSV and it is generally assumed that PRRSV infection, virus load, and lesions are enhanced by this bacterium.
Actinobacillus pleureopneumoniae (APP). There are several strains of APP with significant differences in virulence between strains. Affected lungs have variable degrees of dark-red-purple consolidation with fibrin on the pleural surface. Lesions are often most severe and apparent on the dorsal and caudal portion of the lung. Characteristic microscopic lesions include severe pulmonary hemorrhage and necrosis and bacteria associated with damaged neutrophils. Previous PRRSV infection is not necessary for APP to induce its full spectrum of lesions under experimental conditions; however, evidence from the field suggests APP outbreaks are more severe and common in PRRSV-infected herds.
6. Summary
PRRSV continues to be the most important respiratory pathogen in the global pig population. Evidence from experimental studies in pigs singularly infected with PRRSV suggests that PRRSV by itself may induce mild-to-severe respiratory disease but rarely results in mortality. Morbidity is more severe and mortality occurs with concurrent infections by a variety of other pathogens. In the U.S., the combination of pathogens that caused very severe mortality in 2004-2007 usually included PRRSV, PCV2 and M. hyopneumoniae. Controlling PCV2 through the use of PCV2 vaccines had a major impact on decreasing the severity of PRDC.