Porcine circovirus type 2 (PCV2) is a small, single-stranded, circular-arranged DNA virus associated with a range of disease manifestations in growing pigs collectively known as porcine cirocovirus associated diseases (PCVAD). The main PCV2 proteins include proteins associated with replication encoded by open reading frame 1 (ORF1) and the capsid protein encoded by ORF2. Based on sequence identity in ORF2, PCV2 can be further subdivided into different genotypes. These PCV2 genotypes have been designated consecutively based on the time of first identification with lower case letters (a, b, c, d and e; Fig. 1). PCV2a was long the predominant strain in the global pig population and is also the basis of all major commercially available PCV2 vaccines. Between 2000 and 2005, a genotype shift towards PCV2b occurred. Today, PCV2b is the most commonly identified strain in pigs.
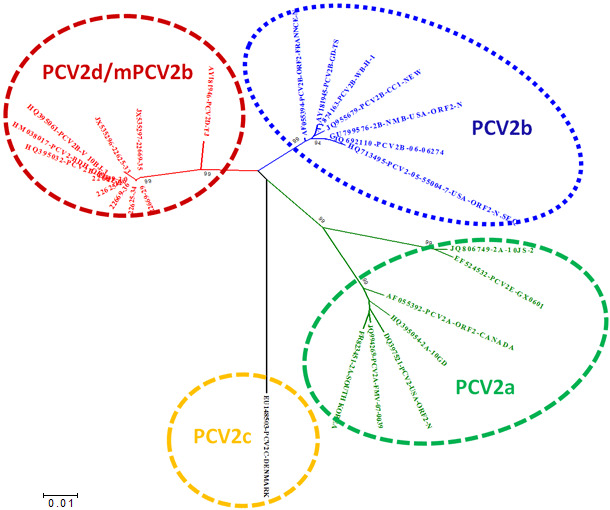

Fig. 1. Relationship of the main PCV2 genotypes based on ORF2 sequence comparison.
During 2012 PCV2 variants or mutants (mPCV2) within the PCV2d cluster appeared in the USA and were frequently associated with PCV-associated diseases (PCVAD). The hallmark difference of these novel strains was an additional amino acid in ORF2 compared to traditional PCV2a and PCV2b strains (234 amino acids instead of 233). However, there are also numerous other amino acid differences throughout ORF2 between mPCV2, PCV2a and PCV2b which are of unknown relevance at this point. Although mPCV2b has only recently (2012) been identified in North America, similar viruses have been present in Asia and Europe for many years indicating that this PCV2 genotype is not novel. Interestingly, in geographic regions where PCV2 sequencing is conducted on a regular basis it appears that the prevalence of mPCV2 strains continues to increase in recent years perhaps indicating some selective advantage of this virus group.
Cross-protection of PCV2a and PCV2b is well documented and is further supported by field evidence of excellent control of PCVAD in herds using commercial vaccines. With the emergence of mPCV2, producers and veterinarians are increasingly reporting and further investigating what they feel may be “vaccine failures” manifest as re-emergence of PCVAD in vaccinated herds. It is well recognized that “vaccine failures” can be caused by several things including failure to comply with recommended procedures while vaccinating the pig, improper vaccine storage and handling conditions, use of reduced dose and other procedural issues. The possibility of lack of cross-protection of PCV2a-based vaccines against mPCV2 is also now of major concern. To address these concerns, a small pilot study evaluating the efficacy of a commercial PCV2a-based vaccine and an experimental mPCV2-based vaccine against mPCV2 challenge was conducted. Specifically, cesarean-derived, colostrum-deprived pigs were utilized, vaccinated twice in a 2-week interval, and were challenged with a tissue homogenate containing mPCV2 and PRRSV two weeks after booster vaccination. A group of pigs was left unvaccinated and served as positive control. Vaccinated and non-vaccinated pigs were comingled and kept in the same pens. After challenge, mPCV2 was identified at low levels in individual pigs in the group vaccinated with the commercial vaccine but was not identified in any of the mPCV2-vaccinated pigs. However, essentially all non-vaccinated positive controls pigs developed high levels of PCV2 viremia and subsequent clinical manifestation of PCVAD. The results of this small pilot study and also the results of a subsequent study in conventional pigs with similar outcomes would certainly indicate that commercial vaccines are capable of protecting against mPCV2 and are encouraging for the pig industry further reassuring that current tools to combat PCVAD are effective.