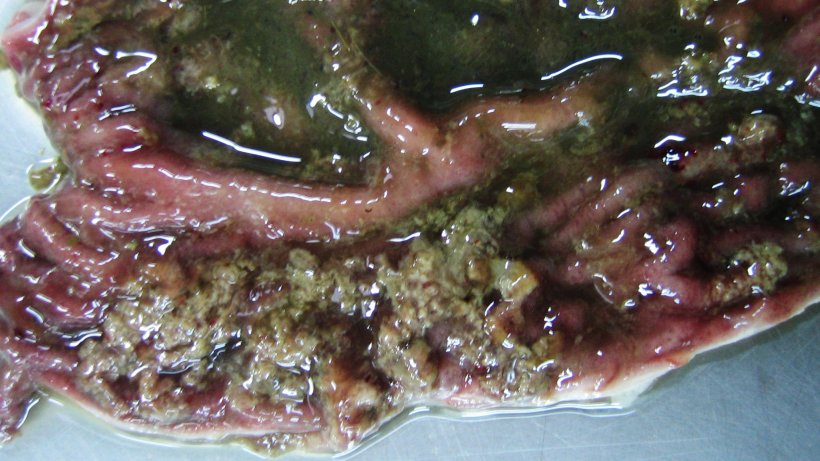
Brachyspira hyodysenteriae is the primary etiologic agent of swine dysentery. Two other species of Brachyspira had been characterized as also primary causative agents of swine dysentery, B. suanatina (Rasback et al, 2007) and B. hampsonii, genomovars I, II and III (Mirakjar et al, 2016). The disease causes severe mucohemorrhagic diarrhea, and thereby severe economic loses to swine production. Gross lesions include variable amounts of mucosal thickening, multifocal hemorrhage, necrosis associated fibrinous exudate, and variable but often excessive luminal mucus in the large intestine (Hampson, 2012). Swine dysentery has a worldwide distribution, occurring mainly in the regions with the highest density of pig production (Calderaro et al., 2001). Although the disease has been absent in North American swine herds in the last 20 years, since 2008, mainly due to management of production systems, several cases have been reported in the United States and Canada (Chander et al., 2012). Recent studies describe the occurrence in Europe (Dors et al., 2015; Löbert et al., 2016) and Asia (Kajiwara et al., 2016). In Brazil, swine dysentery was described from 1980s to 1990s in sporadic reports in small herds, in very few epidemiological studies or associated to the optimization of diagnostic techniques (Barcellos et al., 2000). Since 2010, SD outbreaks have been reported in several Brazilian states, located in the regions that concentrate the largest pig production, causing significant economic losses (Daniel and Guedes, 2013).

There have been some reports of B. hyodysenteriae strains of low pathogenicity, which colonizes but do not induce clinical disease (Lyson et al, 1982; Thomson et al, 2001). There is no commercial vaccine available yet against B. hyodysenteriae, despite enormous effort from the scientific community (Song et al, 2009; Jiang et al, 2014; Singh et al, 2015). There are several explanations for these two facts, but one of the most relevant is the huge genetic diversity observed within B. hyodysenteriae species. Many pathogenic factors are speculated as important for manifestation of virulence. For instance, hemolytic activity is often described as a major one. Mahu et al (2016) sequenced seven hemolysis associated genes of B. hyodysenteriae strains isolated from pigs with mild to severe mucohemorrhagic diarrhea. One weakly hemolytic strain showed sequences changes in five of these genes. Recently, however, during a routine screening of fecal samples from a multiplier herd we faced contradictory results. This specific herd did not use any antimicrobial drugs and had no history of swine dysentery, however, during the routine screening of the herd there were some B. hyodysenteriae PCR positive animals and finally we were successful in isolating the bacterium. Preliminary results of a challenge trial with this isolate, comparing to a known pathogenic strain, demonstrated clear low pathogenicity of the first one (Sato et al, 2016). It is important to mention that this low pathogenic strain was strongly hemolytic in vitro. These findings weaken the assumption of hemolytic genes as being important markers for pathogenicity for B. hyodysenteriae strains.

Recent studies (Song et al, 2015; Hampson et al, 2016) demonstrate the effort to develop a serological test using recombinant proteins mainly to tackle the problem of B. hyodysenteriae subclinically infected herds. This could be a future important tool to detect B. hyodysenteriae positive herds with no clinical signs of swine dysentery. However, genetic variation and significant differences in antigenic proteins are significant limitations for the development of a global applicable serology test.
Anyhow, from the practical point of view, how can a veterinarian deal with a multiplier herd or any commercial herd positive for a low pathogenic B. hyodysenteriae? First, we would be assuming it is a low pathogenic strain only based on the positivity in the diagnostic tests, such as PCR, qPCR, bacterial isolation and/or maybe serology, this last one in the future, associated with the absence of clinical signs. Remember, there is no specific marker for pathogenicity yet! Would this strain become pathogenic when introduced in a commercial herd with problems related to pig flow, hygiene, pig density, etc…? Should multiplier herds be screened routinely by molecular or bacteriological tests in fecal samples for B. hyodysenteriae? Or just perform strict routine diarrhea clinical evaluations in the herd and collect any suspicious fecal material for diagnostic work? Should the herd be closed until getting the results back? At this point, there are very many questions remaining to be answered regarding low pathogenic strains of B. hyodysenteriae, and the great majority of them impact directly the daily routine of veterinary, mainly those involved with health assurance of multiplying herds. There is urging demand for a better understanding of low pathogenic B. hyodysenteriae strains. Anyhow, until we have better answers, strict routine clinical evaluations followed by sample collection of any suspicious case and closing the herd until receiving the results has been the more reasonable protocol.






