Clostridium perfringens type A is part of the normal lower small intestine and colon microbiota of pigs and many other animal species. The diagnosis of C. perfringens type A has increased in neonatal piglet diarrhea cases in the last decade. However, the real importance of this bacterium as a primary agent of diarrhea in suckling piglets is not fully known. The main challenge for the correct interpretation of C. perfringens type A role in cases of neonatal diarrhea has been the complexity to reach a conclusive diagnosis. This aspect will be the focus of the present discussion.
The literature describes the clinical presentation of C. perfringens type A cases as piglets developing creamy or pasty diarrhea after two days of birth that could last for up to five days. Feces can sometimes become mucoid. Mortality is not usually observed, but affected animals tend to fallback comparing to unaffected peers, and consequently compromise the weaning weight. There is no remarkable finding indicative of C. perfringens type A at necropsy, what would possibly differentiate it from other possible enteropathogens found in this age animals. Histologically, superficial villous tip necrosis associated with fibrin accumulation might be present. This lesion associated with heavy colonization of bacillary bacterium in close association with injured enterocytes would favor C. perfringens type A diagnosis (Fig. 1). However, these findings are rarely seen. Most often, no microscopic lesion is observed, what raised the speculation that this bacterium infection might cause secretory diarrhea. This possibility was never confirmed.

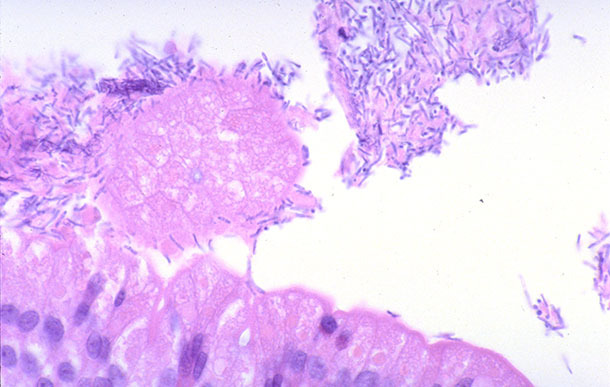
Figure 1. Small intestine of a piglet with diarrhea associated with Clostridium perfringens type A infection. Large amount of bacillary bacteria in close contact with enterocytes (100x). Source: Prof. James Collins, UMN-USA
Gram stain, aspect of bacterium colonies and beta hemolysis in blood agar are the same among all C. perfringens types. The only way to differentiate the five types of C. perfringens, A to E, is based on the production of four major toxins: alpha, beta, epsilon and iota. There are also other two important toxins, enterotoxin and beta 2, that have to be mentioned. All types of C. perfringens can be found in piglet gastrointestinal tract, but only types A and C are considered pathogenic for this age of pigs. There is a very high correlation between the detection of the coding genes of these toxins, cpa, cpb, etx, ia, cpe and cpb2 (Tab. 1) respectively, and their expression, meaning that the PCR detection of these genes would represent the same as the detection of the toxin in the feces. All C. perfringens types have the cpa gene. The detection of cpa and cpb would defined the bacteria as type C, while the detection only of cpa would define the bacteria as type A.
Table 1. Definition of the Clostridium perfringens types based on the production of toxins
| C. perfringens type | Alpha | Beta | Epsilon | Iota | Enterotoxin | Beta 2 |
| A | X | X | X | |||
| B | X | X | X | X | ||
| C | X | X | X | X | ||
| D | X | X | X | |||
| E | X | X | X |
Quantification of C. perfringens type A in fecal material used to be a possible indication of association with diarrhea. In other words, heavy growth of this of C. perfringens type A in primary isolation attempts would strongly indicate its involvement with diarrhea. However, different studies have demonstrated that there is no sufficient evidence suggesting that this quantification could be used as an assay for diagnosing C. perfringens type A infection associated with diarrhea in piglets.
As C. perfringens type A is part of the normal intestinal microbiota of piglets, for many years, the production of beta 2 toxin or the presence of the cpb2 gene by some C. perfringens type A strains thought to be the definitive marker for pathogenicity. As a result, the definitive laboratory diagnosis was based on the detection of beta 2 toxin in fecal material by ELISA or isolation of the agent and typification of the colonies by a multiplex PCR for the presence of cpa and cpb2 genes. However, at least three different studies, including one from our group, have shown that multiplex PCR testing on C. perfringens colonies isolated from diarrheic and normal piglets detected more cpb2 positive C. perfringens type A obtained from normal than sick piglets. Unfortunately, these findings clearly demonstrate there is no specific assay available that can detect or identify pathogenic strains of C. perfringens type A. I truly believe some strains of this bacterium are pathogenic for piglets and can cause diarrhea and reduced growth rate; however, we do not know yet how to identify them! Of course, this condition imposes a tremendous challenge for controlling the disease, as the final diagnosis is based only on the “last possible agent remain for testing”. Much more work has to be done in order to define a possible marker for pathogenicity of C. perfringens type A.




