Oral fluid results from before, at and after start of respiratory symptoms
This poster by N. Wertenbroek was presented at the ESPHM 2018. The different pathogens investigated showed different dynamics. Vaccinations would be also playing a role in this fact. Clearly oral fluids provide a useful tool to find useful information on pathogen dynamics.
PRDC (Porcine Respiratoiry Disease Complex) diagnostics can be easily performed using ropes for Oral Fluids (OFs) as testing material. It is a relative cheap, easy and non-invasive method to collect saliva samples in pens with clinical problems. Combined with PCR it gives more insight in pathogens that are present at the time of clinical signs.
In the period of 2015 and April 2017 samples were collected in farms with respiratory problems in nursery as well as fattening pigs. Ropes were placed at 3 age groups at sampling date:
- “T 0” was the age group were the onset of acute cough was present;
- ”T -2” was the group 2–4 weeks younger than the acute coughing group
- “T +2” was 2–4 weeks older than the acute coughing group.
The age of pigs sampled ranged from weaned pigs till slaughter, depending on the onset of the clinical signs.
Oral fluid samples were collected with ropes, which were placed in the pens for 20–30 minutes1 . Samples were shipped overnight by carrier to the diagnostic lab. The samples were analyzed by a multiplex PCR (IVD-GmbH lab Hannover, Germany) for PRRS, Influenza, PCV2 and Mycoplasma hyopneumoniae. Results were reported as negative or positive for the respective pathogen.
RESULTS
In total 198 samples were collected from 31 farms. The results of the samples were assorted into 5 age groups: 4–6 weeks, 7–9 weeks, 10–13 weeks, 14–18 weeks and 19–24 weeks of age. Figure 1 shows the prevalence for the different pathogens at the different time points before, at and after the start of the cough.
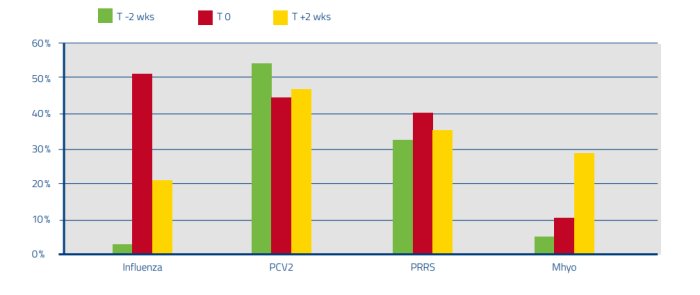
Fig 1: Percentage positive oral fluids at the 3 different time points (before (T-2), at (T0) and after start of cough (T+2))
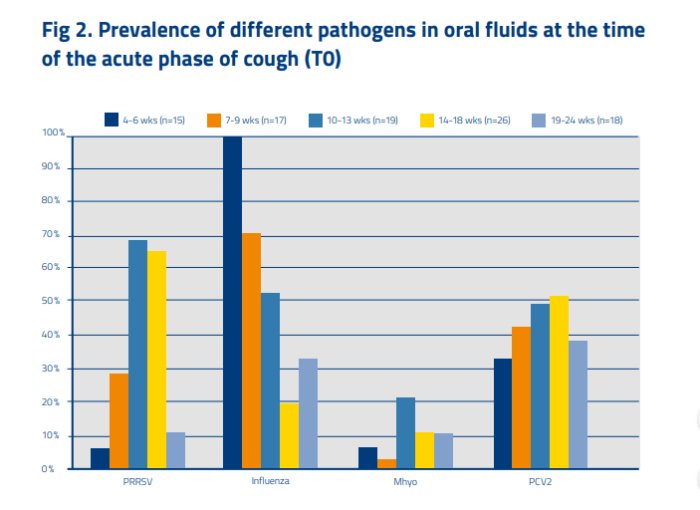
In total, 95 OFs were collected from acutely coughing (T0) pig groups with the majority at the age of 14–18 weeks (n = 26). Figure 2 gives an overview of pathogens detected in acutely coughing pig groups of the different age groups.
DISCUSSION AND CONCLUSION
Different dynamics were found for the 4 investigated pathogens. In acute cough, Influenza (0 % of pigs vaccinated for SIV) has the highest prevalence, indicating to be the primary pathogen especially in the nursery. PRRS is most prevalent in the first 2 months of the finishing phase and was detected more often when cough was present but had also high detection rates at T-2 and T+2. This less clear indication as a primary pathogen may be due to the fact that 36 % of herds had piglets vaccinated. However, in T0 PRRS was present with lower Ct values compared to T-2 and T+2 (data not shown), indicating higher amount of virus particles at acute cough. Mycoplasma is more a secondary agent, and an extender of the cough. Because M hyo has by far the highest prevalence in the T+2 compared to its T0 and T-2, it is at least in this study not the classic ‘door opener’. It can be assumed that this is partly due to the high vaccination rate (pigs in 75 % of farms vaccinated). For PCV2, no real differences were found in prevalence in T-2, T0 and T+2 which is probably due to the high vaccination rate (96 %).
This poster by N. Wertenbroek was presented at the ESPHM 2018.
Contact:
Contact us using the following form.

