Porcine circovirus 2 (PCV2), the cause of porcine circovirus-associated disease (PCVAD), is widespread in swine farms throughout the world.
Infection of pigs is thought to occur at 10-15 weeks of age, when viremia appears, and nearly all finishing pigs are infected with PCV2. Vaccination around the time of weaning is effective in preventing PCVAD and in reducing the level of PCV2 viremia, but it does not eliminate infection. Since vaccination does not eliminate infection and finishing pigs are infected with PCV2, it suggests that gilts and sows might be infected and able to transmit PCV2 to piglets in utero or after birth.

Thus, a study was performed in midwestern US sow farms to evaluate PCV2 exposure, transmission dynamics, and immunological impacts at the time of farrowing.We determined the PCV2 virological and immunological status of sows, pre-suckling piglets, and the farrowing environment.
Results
PCV2 was present in sows on all 6 farms, with 73% of sows being viremic. Viremia levels were highly variable among sows and among farms, and was not influenced by parity or sow vaccination status. Even in sows that were PCR-negative in serum, PCV2 was still observed in oral fluids, colostrum, or feces. Overall, 58 of 59 sows tested sample positive in one or more test materials (Figure 1).
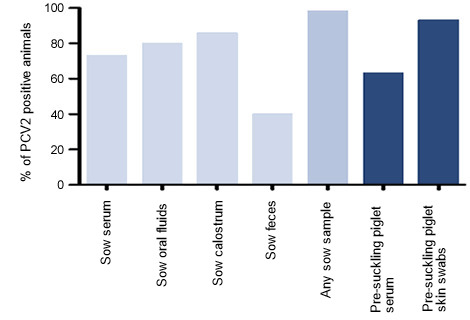
Figure 1. Percent of PCV2 DNA positive animals
All sows, regardless of PCV2 viremia status, had high levels of PCV2-specific IgG, with high anti-capsid antibody levels in all sows on vaccinated farms. However, viremia and antibody levels in serum in individual sows were not correlated. PCV2-specific IgG levels in colostrum were present at high levels, especially in vaccinated sows, and were correlated with serum antibody levels. Anti-PCV2 antibodies were also observed in oral fluids, but at lower levels (Figure 2).
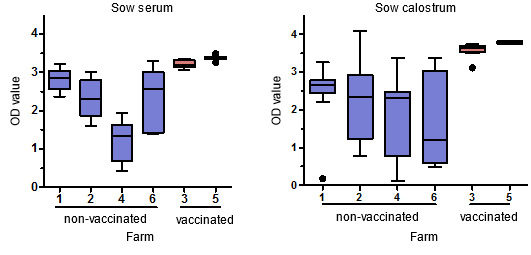
Figure 2. PCV2-specific IgG antibody levels measured by ELISA in sow serum and colostrum.Sow serum samples are diluted 1:50 and colostrum samples are diluted 1:5000.
Pre-suckling piglet serum was PCV2-positive in 63% of 281 piglets, indicating widespread in utero infection. Piglets from non-viremic sows usually had low or undetectable levels of viremia, whereas piglets from viremic sows gave a wide range of viral DNA levels, suggesting that sows with low levels of infection are less likely to pass virus to piglets in utero. Interestingly, axillary skin swabs from pre-suckling piglets were highly positive,with PCV2 levels correlating to viral levels in sow serum and uterine fluid, confirming that piglets are exposed to and infected with PCV2 in utero or at the time of birth (Figure 1).
Widespread shedding from sow secretory and excretory compartments suggests that the piglet-rearing environment was heavily contaminated with PCV2. Not surprisingly, PCV2 DNA was readily detected throughout the piglet environment irrespective of sow vaccination status. High levels of PCV2 DNA were readily detected with or without high pressure washing, and were decreased but not eliminated after disinfection. Live PCV2 was cultured from biological samples, but attempts to isolate PCV2 virus from PCR-positive environmental samples were not successful.
Conclusions and implications

PCV2 infection appears to persist for the lifetime of pigs, since virus was present in sows of all parities, even in the presence of a robust antibody response. Piglets are readily infected with PCV2 in utero and are under constant exposure to PCV2 that is present in colostrum, milk, secretions, and on the skin of sows and in the farrowing environment. Anti-PCV2 antibodies are present in colostrum and oral fluids, but they do not prevent transmission to piglets or control the infection in sows. Overall, a large majority of sows are PCV2-positive and piglets are born PCV2-positive. It is possible that maternal antibodies control infection, since there is no clinical disease in sows or piglets.
The findings indicate that infection of pigs occurs at birth, but that maternal immunity suppresses viremic infection in growing pigs until immunity wanes around 10 weeks of age, when a hidden infection re-appears. The mechanism by which vaccination prevents PCVAD is not known, and is surprising since natural immunity is not effective. The knowledge that infection occurs early in life is important for development of experimental immunity models that usually assume that vaccination precedes infection. In the case of PCV2, it appears that vaccination is occurring in animals that are already infected.


