Introduction
The farm is a commercial 400 sow birth to bacon unit in Northern Ireland.

The unit is negative to Swine Influenza and PRRS (Porcine Reproductive and Respiratory Syndrome).
The vaccination protocol operated is illustrated in Table 1.
Table 1. Vaccination programme
| VACCINE | GILTS | SOWS | BOARS | PIGLETS |
| Parvovirus | 2 ml injection @ selection | 2 ml injection 2 weeks post farrowing | ||
| Erysipelas | 2 ml injection @ selection and 2 weeks pre service | 2 ml injection 2 weeks post farrowing | 2 ml injection twice a year | |
| Colibacillosis | 2 ml injection 6 and 2 weeks pre farrowing | 2 ml injection 2 weeks pre farrowing | ||
| PCV-2 | 2 ml injection 3 weeks pre service and 3 weeks pre farrowing | 2 ml injection 3 weeks pre farrowing | 2 ml injection twice a year | |
| Enzootic Pneumonia | 1 ml @ 7 days and @ weaning |
Gilts are homebred in the unit. Gilts and sows are served with Artificial Insemination from a single source.
History
The farmer contacted the veterinary surgeon concerning an increase of subcutaneous haemorrhages and bruises in the skin of several piglets in eight different litters. No deaths have yet been attributed to the problem.
The unit had experienced this problem during the previous 3 years but not to the same extent as now. The clinical signs appear suddenly in a few litters and then disappear over periods of time.
Investigation
Clinical Investigation
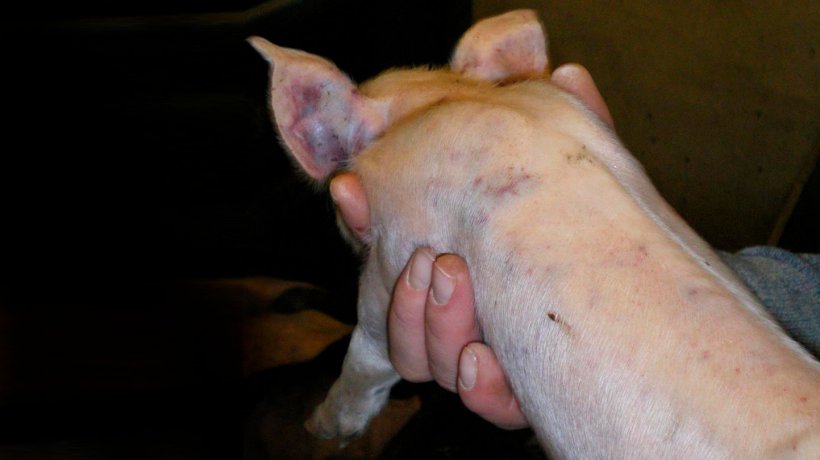
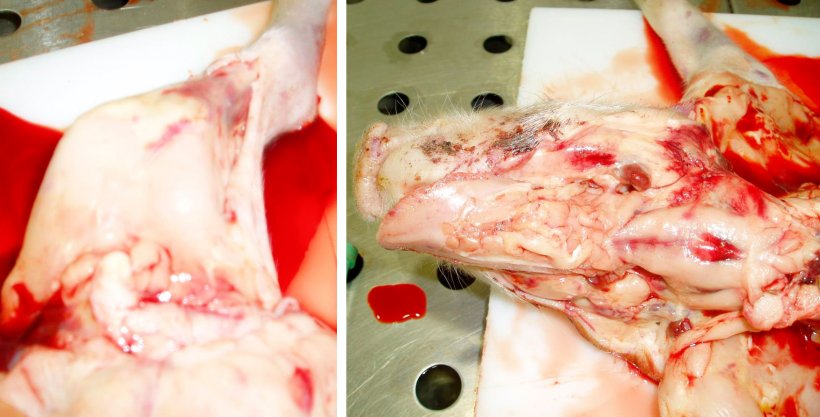
During a farm visit, 28 piglets from 8 different litters had widespread sub-epithelial haemorrhages (Figures 1, 2, 3, 4 & 5). Piglets developed the clinical signs from 7 days of age, after being born normal. Affected piglets continued sucking and they did not appear stunted in any way. Not all the piglets of the affected litters had external clinical signs.

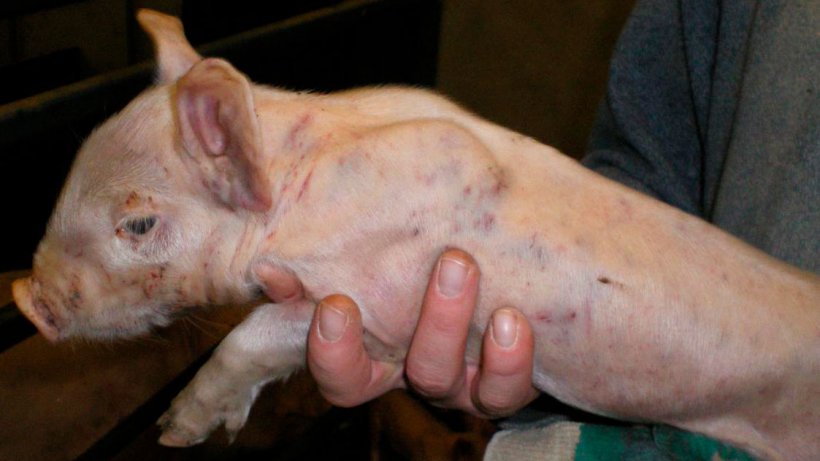
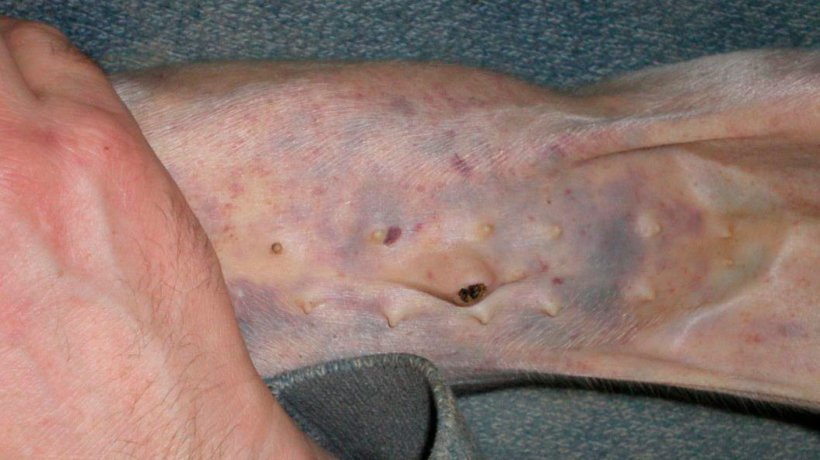
Piglets were being tail-docked at the time of the veterinary farm visit. Bleeding from the tail was profuse and there was a time delay in clotting (Figures 1 & 2).
No fever was detected when taking rectal temperature from 10 affected piglets.
Laboratory Investigations
A recently dead, along with an affected live 7 day old piglet, were collect and submitted to the laboratory. A heparinised blood sample was requested for collection in the laboratory for haematology. The results of laboratory investigations are summarized in Table 2.
Haematology and biochemistry investigations could not be performed as a consequence of the bloods, collected in the laboratory, being clotted.
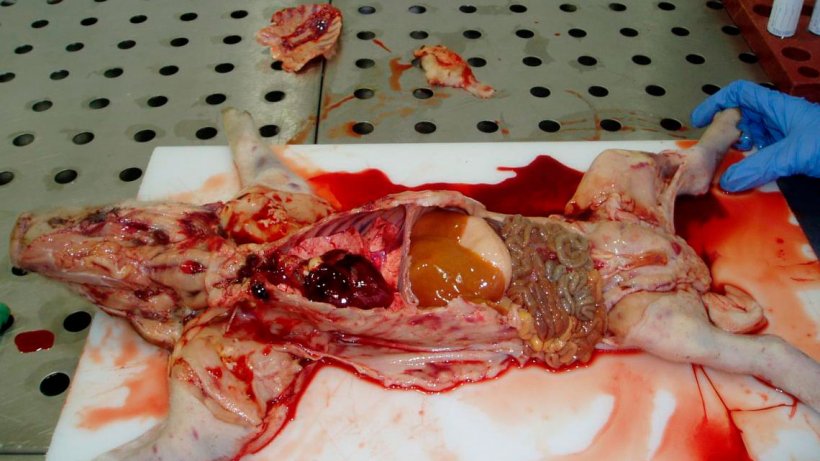
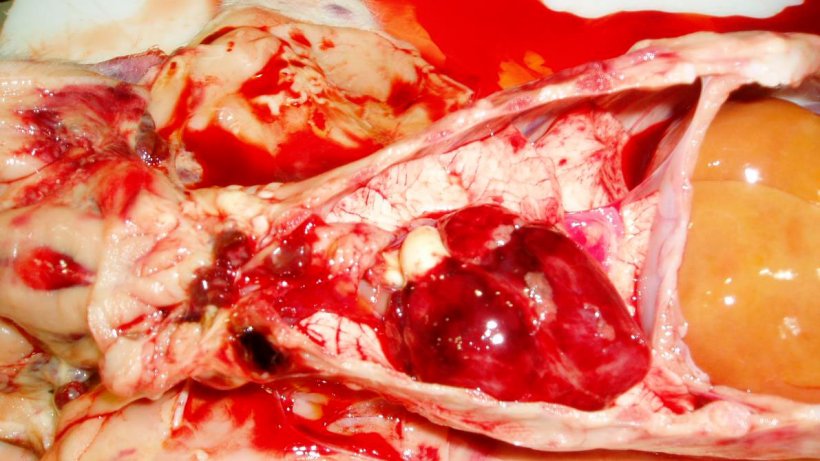
Photographic post-mortem evidence is shown in Figures 6, 7, 8 & 9.
Table 2. Laboratory results from the two 7 day old piglets.
| POST-MORTEM | HISTOLOGY | IMMUNOFLUORESCENCE | BACTERIOLOGY | |
| PIGLET 1 (Dead) | • Widespread superficial sub-epithelial haemorrhages with some petechiation in carcase. • Blood thin and watery. • Some petechial haemorrhages in heart. • Kidney pale and with petechial haemorrhages. • Liver pale and jaundice. |
• No significant abnormalities detected. | • Negative to CSFV and PCV-2. | • No significant bacteria isolated. |
| PIGLET 2 (Live) | • Widespread sub-epithelial haemorrhages in carcase. • Blood thin and watery. • Petechial haemorrhages in kidney and lung. |
• No significant abnormalities detected. | • Negative to CSFV and PCV-2. | • No significant bacteria isolated. |
CSFV = Classical Swine Fever Virus
PCV-2 = Porcine Circovirus type 2
Differential diagnosis
Based on the clinical and laboratory investigations, a list of differential diagnosis was prepared as follows:
- Thrombocytopaenic purpura
- Iron deficiency
- Vitamin K deficiency (Rodenticide Poisoning)
- Classical Swine Fever (CSFV)
- Porcine Circovirosis (PCV-2)
Viral agents, such as CSFV and PCV-2, were ruled out on the basis of lack of clinical signs matching with these diseases and negative immunofluorescence test results.
Iron deficiency was ruled out on the basis of routine iron dextran injections provided to all piglets intramuscularly at 2 days of life and widespread sub-epithelial haemorrhages.
Vitamin K deficiency (Rodenticide Poisoning) could not be excluded of the differential diagnosis without haematology. As referred to 4.2., the blood sample collected clotted and no haematology/biochemistry test could be performed. Rat poison was not placed in the farrowing houses.
A diagnosis of thrombocytopaenic purpura was indicated based on the history and consistent clinical and laboratory investigations.
Control programme
Affected litters were cross-fostered to other sows.
Sows that produced affected litters were recommended to be culled or mated with a different boar.
Response to the control programme
Some of the cross-fostered affected piglets died within few days.
No further cases have been described by the farmer since.
Discussion
Thrombocytopaenic purpura is a type II hypersensitivity (or cytotoxic-type hypersensitivity) in which autoantibodies have been formed against thrombocytes. This results in their depletion and bleeding diathesis as its associated clinical sign. These autoantibodies may arise from blood transfusions, from the use of vaccines that contain blood products, or in multiparous sows that develop antibody against the alloantigens shared by the sire and the foetus. In the latter case, one would not expect clinical signs to appear in the sow since she would only produce antibody against cell-surface allotype antigens that are not found on her cells (Straw et al., 1999). Unlike human babies and puppies, no antibodies are transferred through the placenta from the sow to her piglets before birth. Piglets are born in a vulnerable state without any humoral or mucosal associated antibodies and no acquired cell-mediated immunity (Muirhead et al., 1997a). This would explain why pregnancy is not stopped. The ingestion of these autoantibodies in the colostrum and their absorption by the piglet results in a fall in the circulating levels of thrombocytes to about 200x109/l at 2 days of age (normally higher, 250 – 300x109/l). Recovery takes place to 400x109/l at 5 days and from the 9th day onwards declines to a level of 50x109/l when purpura appears. Megakaryocytes and other cells in the bone marrow are affected and replacement of circulating thrombocytes does not occur (Taylor, 1995).
Blood transfusions and vaccines containing blood products, such as Crystal Violet Swine Fever Vaccine (Smith et al., 1990), were not used in this unit. Therefore, the only option left was multiparous sows that had developed antibody against the alloantigens shared by the sire and the foetus. Affected litters were coming from a Large White boar. All the sows that had produced an affected litter had more than one parity. According to Taylor (1995), in order for the disease to occur, the thrombocyte type of the boar must differ from that of the sow and she must have had at least one previous litter to a boar of that thrombocyte type. It appeared that the affected sows had been A.I. from this boar in a previous litter in this case. Taylor (1995) states that this problem is most frequent in Large White and Landrace pigs and is particularly common when a Large White boar is used on a Landrace sow. In this case since the A.I. used was from a Large White boar and the mated sows were pure Landrace or Landrace x Large White crossed. For this reason, a change of A.I. was recommended which consequently improved the situation without the need to cull the sows or the boar. Unfortunately, thrombocyte count could not be carried out due to the clotting of the blood sample collected in the laboratory. Another alternative would have been collecting the blood on-farm. However, no further cases have since occurred.
Not all piglets of the affected litters showed clinical signs. According to Straw et al. (1999) and Taylor (1995), the random effect of the condition in a litter appears to be related to growth and the ingestion of colostrum, well-grown piglets having sucked more colostrum being most severely affected. This was also true with these piglets since all affected animals had good body condition, were well-grown and did not loose appetite.
Pigs affected by this condition appear normal at birth (Straw et al., 1999). Different authors have reported different timings for first appearance of this condition, 7 – 21 days of age (Muirhead et al., 1997b), 10 – 20 days of age (Straw et al., 1999) or 14 – 30 days of age with a lesser peak at 3 days of age (Taylor, 1995). In this case, disease appeared from 7 days of age, as Muirhead et al. (1997b) states.
Death may occur without clinical signs with good pigs found dead (Muirhead et al., 1997b; Taylor, 1995). If you look closely at the skin of these dead pigs, there should be haemorrhages wherever there has been bruising, teeth marks or trauma (Muirhead et al., 1997b). In this case was not possible to correlate sudden deaths caused by thrombocytopaenic purpura and other causes of death, such as overlaid. However, affected piglets are often pale and a blotchy, purple skin, haemorrhage, mainly on the belly, and vivid scratch marks may be seen. Mucous membranes are usually pale and there is no fever (Straw et al., 1999; Taylor, 1995). Haemorrhages of varying sizes are seen in the epicardium, myocardium, pleura, joints and skeletal muscles. All lymph nodes are engorged with blood (Muirhead et al., 1997b; Taylor, 1995). All these features were consistent with the clinical picture seen in this case.
Castration during the period of thrombocytopaenic purpura may greatly increase the death rate (Straw et al., 1999). Castration was not carried out in this unit. Nevertheless, bleeding was profuse and clotting time was delayed after tail docking, as stated in the Clinical Investigation.
The incidence of this condition is low, less than 1% according to Taylor, 1995.
Diagnosis is done by the characteristic clinical and post-mortem picture (Muirhead et al., 1997b; Taylor, 1995). Diagnosis may be confirmed by platelet counts (Taylor, 1995). All these procedures were followed. However, platelet counts could not be performed by the reasons stated in Laboratory Investigations.
There is no known treatment other that good nursing (Muirhead et al., 1997b; Taylor, 1995). The use of vitamin K has not been reported (Taylor, 1995) but it would be interesting to use it in future outbreaks. In the early stages of the disease it is worth while cross-fostering litters to remove exposure to any lingering antibodies in the sow’s milk (Muirhead et al., 1997b), as carried out in this case. Artificial feeding can also be practiced (Taylor, 1995). However, mortality is quite high.
Thrombocytopaenic purpura has been described in other species, such as cattle and sheep as a consequence of bracken poisoning (Barlow, R., 1983). Humans get this condition as a consequence of infectious, autoimmune, malignant or idiopathic aetiology (Wei, A. et al., 2007).
When a sow has produced such a litter, she should be culled or mated with a different boar at the next pregnancy (Muirhead et al., 1997b; Taylor, 1995). The unit took the option of mating the affected sows with a different boar instead of culling them. This cause of action appears to have been successful as no further cases of thrombocytopaenic purpura have been reported since.







