Description of the farm
A newly established farrow-to-finish farm located in south of Poland was populated with 900 hybrid high health status gilts (PCV2 positive). For on-site production of replacement gilts, an internal multiplier herd was created. Dams were vaccinated against erysipelas, neonatal colibacillosis and porcine parvovirus (PPV). Piglets were weaned at 3 weeks of age.

PRRSV outbreak
Periodical serological monitoring showed that for 7 months the herd remained free from PRRSV. Then, reproductive failure was seen in sows. Conception rates dropped from 89.3% to 62%, and stillborn piglets increased from 2.8 to 6.4% (Fig. 1a). Coincident with the reproductive failure, seroconversion to PRRSV was detected, with indirect discriminatory ELISA results indicating infection with European genotype PRRSV.
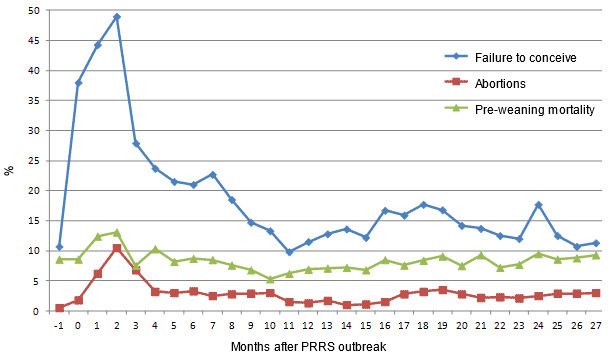
Fig 1.a Selected parameters of the herd performance since the month preceding a PRRS outbreak (-1) until 27 months after the outbreak
PMWS outbreak
One month after the beginning of the outbreak of reproductive failure in sows, acute respiratory disorder and diarrhoea that was non-responsive to antibiotics started in weaned pigs aged 6-8 weeks. The mortality in this group increased from 1.8% to 4.5% at 1 month after these symptoms were noted. Also, the percentage of poor-doing weaners increased in the same period from 4% to 7.9% (Fig. 1b). Post-mortem examination of 4 wasted pigs of about 8 weeks of age revealed enlarged lymph nodes, enlarged livers and enlarged and pale kidneys with generalized cortical petecchiae. Microscopic analysis of lymph nodes revealed different degree of lymphocyte depletion and histiocytic infiltrations in all animals. In situ hybridization (ISH) using a PCV2 specific DNA probe showed abundant presence of PCV2 DNA within these lesions. Together, the histopathological and clinical data fulfilled the PMWS case definition criteria.
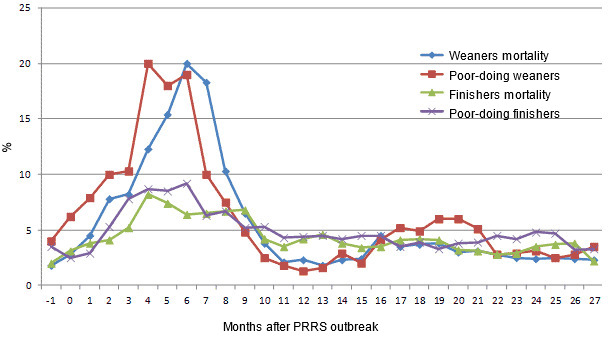
Fig 1.b Selected parameters of the herd performance since the month preceding a PRRS outbreak (-1) until 27 months after the outbreak
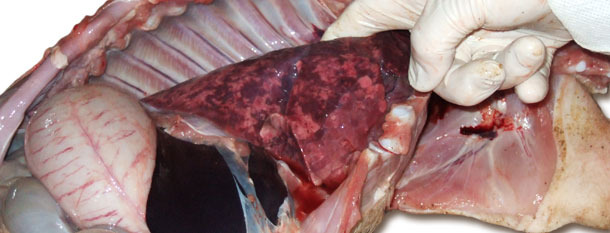
Image 1. Enlarged, non-collapsed, rubbery, patchy lungs correspond to interstitial pneumonia

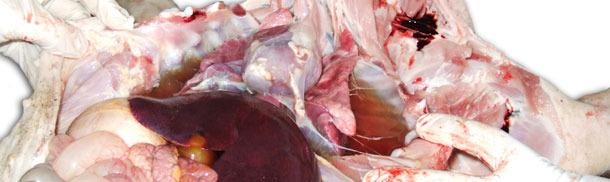
Image 2. Liquid in thoracic cavity
Control measures
Immediately after the detection of the PRRS outbreak, the sow herd and the internal multiplier herd were closed for replacement for three months, in order to control PRRSV circulation. Later, only internally grown PRRSV-seropositive gilts were used for replacement. These gilts were exposed to the local PRRSV strain in fattening units, where they were placed for about three to four months. Before mating, PRRSV seroconversion was verified by serology.
After diagnosis of PMWS the following measures were applied:
- Improvement of environmental conditions (air quality, temperature),
- improvement of feeding of sows and weaned pigs with a feed containing increased level of vitamin E for sows and piglets,
- limited cross-fostering,
- dry disinfection of farrowing units two days after farrowing,
- extension of the lactation period from 21 to 26 days,
- strict all-in, all-out pig flow in nursery and finishing units with rigorous cleaning and disinfection between the batches of pigs,
- separation of sick from healthy pigs, with immediate euthanasia of sick animals, and
- unidirectional work flow for the farm staff, from younger to older pigs, with separate sets of clothing and boots for every room.
Impact of the measures on PRRS
Following the herd closure, abortion rate and pre-weaning mortality remained at increased levels for approximately 4 months. The highest level of reproductive losses was observed 2 months after the PRRS outbreak, when the abortion rate reached 10.5% (Fig. 1a). The conception rate dropped from 90% in the month preceding the PRRS outbreak to 50% three months later. Four months after the outbreak the abortion rate returned to the acceptable level of 3.2% but the conception rate remained decreased. Eight months after the outbreak the conception rate reached 81.5% and remained above this level in the following months. The highest proportion of stillborn piglets (8.2%) was recorded 2 months after the beginning of the PRRS outbreak and remained increased until 7 months after the outbreak. Also, the pre-weaning mortality level increased to 13.1% 2 months after the outbreak and returned to pre-outbreak value within the next 3 months (Fig . 1a).
Impact of the measures on PMWS
Slower improvement was observed regarding the PMWS outbreak. Between 3 and 6 months after the start of the PRRS outbreak, weaner mortality increased from 8.2% to 20%, and the proportion of poor-doing weaners reached 20% (Fig. 1b). During the following 4 months, the PMWS related problems in weaners started to wane and eventually disappeared.
During the acute outbreak of PMWS, also health condition of the fatteners up to 16 week of age was compromised. Mortality in fatteners increased from 3.1% before the start of the PRRS outbreak (month 0) to 8.2% 4 months after the start of the outbreak. Before the PRRS outbreak the proportion of poor-doing fatteners was 2.5% and reached a peak of 9.2%, 4 months after the start of the outbreak (Fig. 1b). Performance in fatteners remained decreased for 10 months after the beginning of the outbreak and returned to normal level after this period.
Results of serological monitoring of PRRSV and PCV2 circulation
Analysis of the serological results of pigs sampled 1 month after the outbreak indicated that PRRSV infection occurred before weaning. Five months later the seroconversion to PRRSV was occurring around the time of weaning and 9 months after the beginning of the PRRS outbreak no antibodies were detected in suckling piglets or weaners. This showed that PRRSV was eliminated from the sow herd and all the piglets were born free from this virus. However, PRRSV seroconversion was still detected in fatteners, showing that PRRSV circulation was maintained in these units.
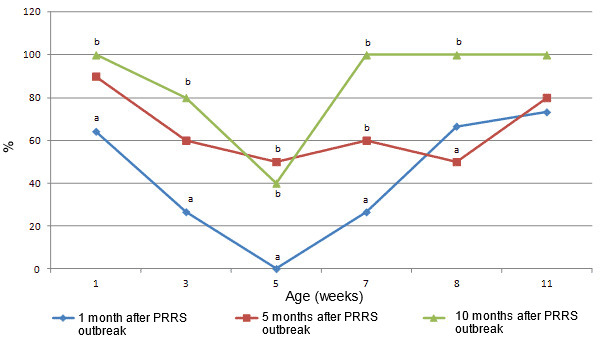
Fig. 2. Seroconversion to PCV2. Proportion of sera positive in IPMA at dilution 1:500 in months 1, 5 and 10 following the PRRS outbreak, in 1, 3, 5, 7, 9 and 11 weeks old pigs. Different letters (a,b) indicate statistically significant differences between proportion of seropositive pigs at 1, 3, 5, 7 and 9 weeks of age (p < 0.05).
Interestingly, no changes were observed in PCV2 circulation between month 1 (the start of the PMWS outbreak), month 5 (the peak of the PMWS outbreak) and month 10 (after the PMWS outbreak). At all these times, the youngest animals showing seroconversion to PCV2 were 7 weeks old. However serological analysis showed that levels of PCV2 antibodies transferred with colostrum to piglets increased with time. Also, seroconversion at 7 weeks of age was increasingly effective in time. While in the first month of the PMWS outbreak only 27% of these pigs was seropositive, 10 months later all seroconverted, indicating improved humoral immune response to infection with PCV2. Thus, these results suggest that both, passive and active seroconversion increased over time and this improvement was correlated with the control of the PMWS outbreak.
Conclusion
In summary, the herd closure and use of internally bred and PRRSV-seropositive replacement gilts, apparently assisted to build up effective immunity leading to elimination of PRRSV in the sow and farrowing units, and the reproductive performance returned to normal 8 months after the PRRS outbreak.
The seronegative gilts (weight 40-50 kg) were placed in fattening units. Periodical serological monitoring of the acclimation efficacy showed that after 1 month about 30% of gilts seroconverted and after 3 months 100% seroconverted. Acclimation was thus done for 4 months, to allow the gilts to completely eliminate PRRSV before mating. Ten months after the first PRRS symptoms, PRRSV circulation (assessed by serological profiles of the herd) was eliminated from weaners and was seen only in the fattening units.
In summary, we believe that in the described case the outbreak of PMWS in piglets appeared to have been triggered by a preceding PRRS outbreak, and resulted low levels of maternal PCV2 antibodies transferred to piglets. Elimination of PRRSV from sows and later from weaners significantly contributed to elimination of the PMWS from the herd.
This case report was published by Stadejek T., Podgorska K., Porowski M., Jabłoński A., Pejsak Z. and can be found at Veterinary Record doi:10.1136/vr.d4840